Centrin depletion causes cyst formation and other ciliopathy-related phenotypes in zebrafish
- PMID: 22142866
- PMCID: PMC3266121
- DOI: 10.4161/cc.10.22.18150
Centrin depletion causes cyst formation and other ciliopathy-related phenotypes in zebrafish
Abstract
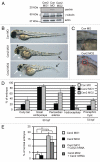
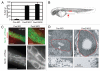
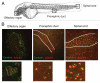
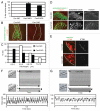
Most bona fide centrosome proteins including centrins, small calcium-binding proteins, participate in spindle function during mitosis and play a role in cilia assembly in non-cycling cells. Although the basic cellular functions of centrins have been studied in lower eukaryotes and vertebrate cells in culture, phenotypes associated with centrin depletion in vertebrates in vivo has not been directly addressed. To test this, we depleted centrin2 in zebrafish and found that it leads to ciliopathy phenotypes including enlarged pronephric tubules and pronephric cysts. Consistent with the ciliopathy phenotypes, cilia defects were observed in differentiated epithelial cells of ciliated organs such as the olfactory bulb and pronephric duct. The organ phenotypes were also accompanied by cell cycle deregulation namely mitotic delay resulting from mitotic defects. Overall, this work demonstrates that centrin2 depletion causes cilia-related disorders in zebrafish. Moreover, given the presence of both cilia and mitotic defects in the affected organs, it suggests that cilia disorders may arise from a combination of these defects.
Figures
Comment in
-
Many defects make a cyst.Cell Cycle. 2012 Jan 1;11(1):16. doi: 10.4161/cc.11.1.18732. Epub 2012 Jan 1. Cell Cycle. 2012. PMID: 22193033 No abstract available.
Similar articles
-
Depletion of Aurora-A in zebrafish causes growth retardation due to mitotic delay and p53-dependent cell death.FEBS J. 2013 Mar;280(6):1518-30. doi: 10.1111/febs.12153. Epub 2013 Feb 24. FEBS J. 2013. PMID: 23351126
-
Lethal giant larvae 2 regulates development of the ciliated organ Kupffer's vesicle.Development. 2013 Apr;140(7):1550-9. doi: 10.1242/dev.087130. Development. 2013. PMID: 23482490 Free PMC article.
-
Centrosomal protein FOR20 is essential for cilia-dependent development in zebrafish embryos.FASEB J. 2019 Mar;33(3):3613-3622. doi: 10.1096/fj.201801235RR. Epub 2018 Nov 26. FASEB J. 2019. PMID: 30475641
-
In vivo cell biology in zebrafish - providing insights into vertebrate development and disease.J Cell Sci. 2014 Feb 1;127(Pt 3):485-95. doi: 10.1242/jcs.140194. J Cell Sci. 2014. PMID: 24481493 Free PMC article. Review.
-
Role of primary cilia in non-dividing and post-mitotic cells.Cell Tissue Res. 2017 Jul;369(1):11-25. doi: 10.1007/s00441-017-2599-7. Epub 2017 Mar 30. Cell Tissue Res. 2017. PMID: 28361305 Free PMC article. Review.
Cited by
-
CCDC15 localizes to the centriole inner scaffold and controls centriole length and integrity.J Cell Biol. 2023 Dec 4;222(12):e202305009. doi: 10.1083/jcb.202305009. Epub 2023 Nov 7. J Cell Biol. 2023. PMID: 37934472 Free PMC article.
-
miR-34/449 miRNAs are required for motile ciliogenesis by repressing cp110.Nature. 2014 Jun 5;510(7503):115-20. doi: 10.1038/nature13413. Nature. 2014. PMID: 24899310 Free PMC article.
-
The two human centrin homologues have similar but distinct functions at Tetrahymena basal bodies.Mol Biol Cell. 2012 Dec;23(24):4766-77. doi: 10.1091/mbc.E12-06-0454. Epub 2012 Oct 19. Mol Biol Cell. 2012. PMID: 23087207 Free PMC article.
-
Centrin2 regulates CP110 removal in primary cilium formation.J Cell Biol. 2015 Mar 16;208(6):693-701. doi: 10.1083/jcb.201411070. Epub 2015 Mar 9. J Cell Biol. 2015. PMID: 25753040 Free PMC article.
-
Centrin-2 (Cetn2) mediated regulation of FGF/FGFR gene expression in Xenopus.Sci Rep. 2015 May 27;5:10283. doi: 10.1038/srep10283. Sci Rep. 2015. PMID: 26014913 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
