Pharmacological and patient-specific response determinants in patients with hospital-acquired pneumonia treated with tigecycline
- PMID: 22143524
- PMCID: PMC3264202
- DOI: 10.1128/AAC.01615-10
Pharmacological and patient-specific response determinants in patients with hospital-acquired pneumonia treated with tigecycline
Abstract
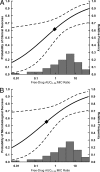
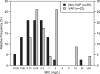
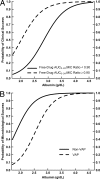
Pharmacokinetic and clinical data from tigecycline-treated patients with hospital-acquired pneumonia (HAP) who were enrolled in a phase 3 clinical trial were integrated in order to evaluate pharmacokinetic-pharmacodynamic (PK-PD) relationships for efficacy. Univariable and multivariable analyses were conducted to identify factors associated with clinical and microbiological responses, based on data from 61 evaluable HAP patients who received tigecycline intravenously as a 100-mg loading dose followed by 50 mg every 12 h for a minimum of 7 days and for whom there were adequate clinical, pharmacokinetic, and response data. The final multivariable logistic regression model for clinical response contained albumin and the ratio of the free-drug area under the concentration-time curve from 0 to 24 h (fAUC(0-24)) to the MIC (fAUC(0-24):MIC ratio). The odds of clinical success were 13.0 times higher for every 1-g/dl increase in albumin (P < 0.001) and 8.42 times higher for patients with fAUC(0-24):MIC ratios of ≥0.9 compared to patients with fAUC(0-24):MIC ratios of <0.9 (P = 0.008). Average model-estimated probabilities of clinical success for the albumin/fAUC(0-24):MIC ratio combinations of <2.6/<0.9, <2.6/≥0.9, ≥2.6/<0.9, and ≥2.6/≥0.9 were 0.21, 0.57, 0.64, and 0.93, respectively. For microbiological response, the final model contained albumin and ventilator-associated pneumonia (VAP) status. The odds of microbiological success were 21.0 times higher for every 1-g/dl increase in albumin (P < 0.001) and 8.59 times higher for patients without VAP compared to those with VAP (P = 0.003). Among the remaining variables evaluated, the MIC had the greatest statistical significance, an observation which was not surprising given the differences in MIC distributions between VAP and non-VAP patients (MIC(50)and MIC(90) values of 0.5 and 0.25 mg/liter versus 16 and 1 mg/liter for VAP versus non-VAP patients, respectively; P = 0.006). These findings demonstrated the impact of pharmacological and patient-specific factors on the clinical and microbiological responses.
Figures
References
-
- Albert A, Anderson JA. 1984. On the existence of maximum-likelihood estimates in logistic regression models. Biometrika 77:1–10
-
- Al-Muhairi SA, Zoubeidi TA, Ellis ME, Safa WF, Joseph J. 2006. Risk factors predicting outcome in patients with pneumonia in Al-Ain, United Arab Emirates. Saudi Med. J. 27:1044–1048 - PubMed
-
- Ambrose PG, Bhavnani SM, Ellis-Grosse EJ, Drusano GL. 2010. Pharmacokinetic-pharmacodynamic considerations in the design of hospital-acquired or ventilator-associated bacterial pneumonia studies: look before you leap! Clin. Infect. Dis. 51(Supp. 1):S103–S110 - PubMed
-
- Ambrose PG. 2008. Use of pharmacokinetics and pharmacodynamics in a failure analysis of community-acquired pneumonia: implications for future clinical trial study design. Clin. Infect. Dis. 47:S225–S231 - PubMed
-
- Anonymous. 2009. FDA asks for more information before sanctioning J &J PRD's drug for hospital-acquired pneumonia. Genet. Engin. Biotechnol. News http://www.genengnews.com/news/bnitem.aspx?name=40760576&source=genwire
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

