Ascorbic acid enhances the cardiac differentiation of induced pluripotent stem cells through promoting the proliferation of cardiac progenitor cells
- PMID: 22143566
- PMCID: PMC3351910
- DOI: 10.1038/cr.2011.195
Ascorbic acid enhances the cardiac differentiation of induced pluripotent stem cells through promoting the proliferation of cardiac progenitor cells
Abstract
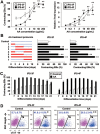
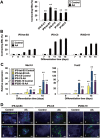
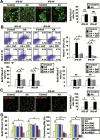
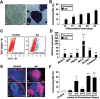
Generation of induced pluripotent stem cells (iPSCs) has opened new avenues for the investigation of heart diseases, drug screening and potential autologous cardiac regeneration. However, their application is hampered by inefficient cardiac differentiation, high interline variability, and poor maturation of iPSC-derived cardiomyocytes (iPS-CMs). To identify efficient inducers for cardiac differentiation and maturation of iPSCs and elucidate the mechanisms, we systematically screened sixteen cardiomyocyte inducers on various murine (m) iPSCs and found that only ascorbic acid (AA) consistently and robustly enhanced the cardiac differentiation of eleven lines including eight without spontaneous cardiogenic potential. We then optimized the treatment conditions and demonstrated that differentiation day 2-6, a period for the specification of cardiac progenitor cells (CPCs), was a critical time for AA to take effect. This was further confirmed by the fact that AA increased the expression of cardiovascular but not mesodermal markers. Noteworthily, AA treatment led to approximately 7.3-fold (miPSCs) and 30.2-fold (human iPSCs) augment in the yield of iPS-CMs. Such effect was attributed to a specific increase in the proliferation of CPCs via the MEK-ERK1/2 pathway by through promoting collagen synthesis. In addition, AA-induced cardiomyocytes showed better sarcomeric organization and enhanced responses of action potentials and calcium transients to β-adrenergic and muscarinic stimulations. These findings demonstrate that AA is a suitable cardiomyocyte inducer for iPSCs to improve cardiac differentiation and maturation simply, universally, and efficiently. These findings also highlight the importance of stimulating CPC proliferation by manipulating extracellular microenvironment in guiding cardiac differentiation of the pluripotent stem cells.
Figures




References
-
- Yu J, Vodyanik MA, Smuga-Otto K, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917–1920. - PubMed
-
- Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. - PubMed
-
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

