Tertiary alkylamines as nucleophiles in substitution reactions at heteroaromatic halide during the synthesis of the highly potent pirinixic acid derivative 2-(4-chloro-6-(2,3-dimethylphenylamino)pyrimidin-2-ylthio)octanoic acid (YS-121)
- PMID: 22143571
- PMCID: PMC6264330
- DOI: 10.3390/molecules161210013
Tertiary alkylamines as nucleophiles in substitution reactions at heteroaromatic halide during the synthesis of the highly potent pirinixic acid derivative 2-(4-chloro-6-(2,3-dimethylphenylamino)pyrimidin-2-ylthio)octanoic acid (YS-121)
Abstract
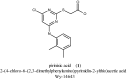
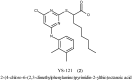
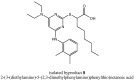
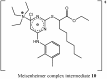
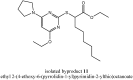
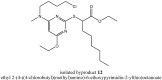
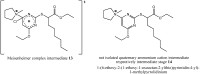
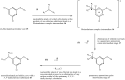
YS-121 [2-(4-chloro-6-(2,3-dimethylphenylamino)pyrimidin-2-ylthio)octanoic acid] is the result of target-oriented structural derivatization of pirinixic acid. It is a potent dual PPARα/γ-agonist, as well as a potent dual 5-LO/mPGES-1-inhibitor. Additionally, recent studies showed an anti-inflammatory efficacy in vivo. Because of its interference with many targets, YS-121 is a promising drug candidate for the treatment of inflammatory diseases. Ongoing preclinical studies will thus necessitate huge amounts of YS-121. To cope with those requirements, we have optimized the synthesis of YS-121. Surprisingly, we isolated and characterized byproducts during the resulting from nucleophilic aromatic substitution reactions by different tertiary alkylamines at a heteroaromatic halide. These amines should actually serve as assisting bases, because of their low nucleophilicity. This astonishing fact was not described in former publications concerning that type of reaction and, therefore, might be useful for further reaction improvement in general. Furthermore, we could develop a proposal for the mechanism of that byproduct formation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Santilli A.A., Scotese A.C., Tomarelli R.M. A potent antihypercholesterolemic agent: [4-chloro-6-(2,3-xylidino)-2-pyrimidinylthio]acetic acid (Wy-14643) Experientia. 1974;30:1110–1111. - PubMed
-
- Reddy J.K., Moody D.E., Azarnoff D.L., Tomarelli R.M. Hepatic effects of some [4-chloro-6-(2,3-xylidino)-2-pyrimidinylthio] acetic acid (WY-14,643) analogs in the mouse. Arch. Int. Pharmacodyn. Ther. 1977;225:51–57. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

