Release of mitochondrial apoptogenic factors and cell death are mediated by CK2 and NADPH oxidase
- PMID: 22146192
- PMCID: PMC3318149
- DOI: 10.1038/jcbfm.2011.176
Release of mitochondrial apoptogenic factors and cell death are mediated by CK2 and NADPH oxidase
Abstract
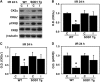
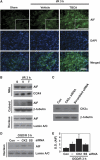
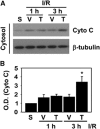
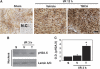
Activation of the NADPH oxidase subunit, NOX2, and increased oxidative stress are associated with neuronal death after cerebral ischemia and reperfusion. Inhibition of NOX2 by casein kinase 2 (CK2) leads to neuronal survival, but the mechanism is unknown. In this study, we show that in copper/zinc-superoxide dismutase transgenic (SOD1 Tg) mice, degradation of CK2α and CK2α' and dephosphorylation of CK2β against oxidative stress were markedly reduced compared with wild-type (WT) mice that underwent middle cerebral artery occlusion. Inhibition of CK2 pharmacologically or by ischemic reperfusion facilitated accumulation of poly(ADP-ribose) polymers, the translocation of apoptosis-inducing factor (AIF), and cytochrome c release from mitochondria after ischemic injury. The eventual enhancement of CK2 inhibition under ischemic injury strongly increased 8-hydroxy-2'-deoxyguanosine and phosphorylation of H2A.X. Furthermore, CK2 inhibition by tetrabromocinnamic acid (TBCA) in SOD1 Tg and gp91 knockout (KO) mice after ischemia reperfusion induced less release of AIF and cytochrome c than in TBCA-treated WT mice. Inhibition of CK2 in gp91 KO mice subjected to ischemia reperfusion did not increase brain infarction compared with TBCA-treated WT mice. These results strongly suggest that NOX2 activation releases reactive oxygen species after CK2 inhibition, triggering release of apoptogenic factors from mitochondria and inducing DNA damage after ischemic brain injury.
Figures


Similar articles
-
CK2 inhibition protects white matter from ischemic injury.Neurosci Lett. 2018 Nov 20;687:37-42. doi: 10.1016/j.neulet.2018.08.021. Epub 2018 Aug 17. Neurosci Lett. 2018. PMID: 30125643 Free PMC article. Review.
-
CK2 is a novel negative regulator of NADPH oxidase and a neuroprotectant in mice after cerebral ischemia.J Neurosci. 2009 Nov 25;29(47):14779-89. doi: 10.1523/JNEUROSCI.4161-09.2009. J Neurosci. 2009. PMID: 19940173 Free PMC article.
-
Inhibition of MiRNA-125b Decreases Cerebral Ischemia/Reperfusion Injury by Targeting CK2α/NADPH Oxidase Signaling.Cell Physiol Biochem. 2018;45(5):1818-1826. doi: 10.1159/000487873. Epub 2018 Feb 28. Cell Physiol Biochem. 2018. PMID: 29510389
-
NOX2 deficiency ameliorates cerebral injury through reduction of complexin II-mediated glutamate excitotoxicity in experimental stroke.Free Radic Biol Med. 2013 Dec;65:942-951. doi: 10.1016/j.freeradbiomed.2013.08.166. Epub 2013 Aug 24. Free Radic Biol Med. 2013. PMID: 23982049
-
Reperfusion and neurovascular dysfunction in stroke: from basic mechanisms to potential strategies for neuroprotection.Mol Neurobiol. 2010 Jun;41(2-3):172-9. doi: 10.1007/s12035-010-8102-z. Epub 2010 Feb 17. Mol Neurobiol. 2010. PMID: 20157789 Free PMC article. Review.
Cited by
-
The Phosphorylated Form of the Histone H2AX (γH2AX) in the Brain from Embryonic Life to Old Age.Molecules. 2021 Nov 27;26(23):7198. doi: 10.3390/molecules26237198. Molecules. 2021. PMID: 34885784 Free PMC article. Review.
-
Ginsenoside Rb1 attenuates oxygen-glucose deprivation-induced apoptosis in SH-SY5Y cells via protection of mitochondria and inhibition of AIF and cytochrome c release.Molecules. 2013 Oct 16;18(10):12777-92. doi: 10.3390/molecules181012777. Molecules. 2013. PMID: 24135936 Free PMC article.
-
Quintessential risk factors: their role in promoting cognitive dysfunction and Alzheimer's disease.Neurochem Res. 2012 Dec;37(12):2627-58. doi: 10.1007/s11064-012-0854-6. Epub 2012 Aug 12. Neurochem Res. 2012. PMID: 22886562 Review.
-
CK2 inhibition protects white matter from ischemic injury.Neurosci Lett. 2018 Nov 20;687:37-42. doi: 10.1016/j.neulet.2018.08.021. Epub 2018 Aug 17. Neurosci Lett. 2018. PMID: 30125643 Free PMC article. Review.
-
PGJ(2) provides prolonged CNS stroke protection by reducing white matter edema.PLoS One. 2012;7(12):e50021. doi: 10.1371/journal.pone.0050021. Epub 2012 Dec 20. PLoS One. 2012. PMID: 23284631 Free PMC article.
References
-
- Ames BN. Endogenous oxidative DNA damage, aging, and cancer. Free Radic Res Commun. 1989;7:121–128. - PubMed
-
- Breusing N, Grune T. Regulation of proteasome-mediated protein degradation during oxidative stress and aging. Biol Chem. 2008;389:203–209. - PubMed
-
- Chan PH. Mitochondrial dysfunction and oxidative stress as determinants of cell death/survival in stroke. Ann NY Acad Sci. 2005;1042:203–209. - PubMed
-
- Chan PH, Epstein CJ, Kinouchi H, Kamii H, Chen SF, Carlson E, Gafni J, Yang G, Reola L. Neuroprotective role of CuZn–superoxide dismutase in ischemic brain damage. Adv Neurol. 1996;71:271–280. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

