Localization of cell wall polysaccharides in normal and compression wood of radiata pine: relationships with lignification and microfibril orientation
- PMID: 22147521
- PMCID: PMC3271756
- DOI: 10.1104/pp.111.184036
Localization of cell wall polysaccharides in normal and compression wood of radiata pine: relationships with lignification and microfibril orientation
Abstract
The distribution of noncellulosic polysaccharides in cell walls of tracheids and xylem parenchyma cells in normal and compression wood of Pinus radiata, was examined to determine the relationships with lignification and cellulose microfibril orientation. Using fluorescence microscopy combined with immunocytochemistry, monoclonal antibodies were used to detect xyloglucan (LM15), β(1,4)-galactan (LM5), heteroxylan (LM10 and LM11), and galactoglucomannan (LM21 and LM22). Lignin and crystalline cellulose were localized on the same sections used for immunocytochemistry by autofluorescence and polarized light microscopy, respectively. Changes in the distribution of noncellulosic polysaccharides between normal and compression wood were associated with changes in lignin distribution. Increased lignification of compression wood secondary walls was associated with novel deposition of β(1,4)-galactan and with reduced amounts of xylan and mannan in the outer S2 (S2L) region of tracheids. Xylan and mannan were detected in all lignified xylem cell types (tracheids, ray tracheids, and thick-walled ray parenchyma) but were not detected in unlignified cell types (thin-walled ray parenchyma and resin canal parenchyma). Mannan was absent from the highly lignified compound middle lamella, but xylan occurred throughout the cell walls of tracheids. Using colocalization measurements, we confirmed that polysaccharides containing galactose, mannose, and xylose have consistent correlations with lignification. Low or unsubstituted xylans were localized in cell wall layers characterized by transverse cellulose microfibril orientation in both normal and compression wood tracheids. Our results support the theory that the assembly of wood cell walls, including lignification and microfibril orientation, may be mediated by changes in the amount and distribution of noncellulosic polysaccharides.
Figures



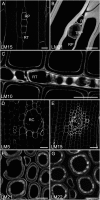
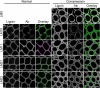

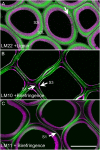
Similar articles
-
Xylem parenchyma cell walls lack a gravitropic response in conifer compression wood.Planta. 2015 Dec;242(6):1413-24. doi: 10.1007/s00425-015-2381-6. Epub 2015 Aug 19. Planta. 2015. PMID: 26287313
-
Distribution of (1->4)-beta-galactans, arabinogalactan proteins, xylans and (1->3)-beta-glucans in tracheid cell walls of softwoods.Tree Physiol. 2010 Jun;30(6):782-93. doi: 10.1093/treephys/tpq021. Epub 2010 Apr 9. Tree Physiol. 2010. PMID: 20382964
-
Comparison of Micropore Distribution in Cell Walls of Softwood and Hardwood Xylem.Plant Physiol. 2018 Nov;178(3):1142-1153. doi: 10.1104/pp.18.00883. Epub 2018 Sep 14. Plant Physiol. 2018. PMID: 30217826 Free PMC article.
-
Covalent interactions between lignin and hemicelluloses in plant secondary cell walls.Curr Opin Biotechnol. 2019 Apr;56:97-104. doi: 10.1016/j.copbio.2018.10.010. Epub 2018 Nov 10. Curr Opin Biotechnol. 2019. PMID: 30423528 Review.
-
Modifying crops to increase cell wall digestibility.Plant Sci. 2012 Apr;185-186:65-77. doi: 10.1016/j.plantsci.2011.10.014. Epub 2011 Oct 25. Plant Sci. 2012. PMID: 22325867 Review.
Cited by
-
Proximal and Distal Parts of Sweetpotato Adventitious Roots Display Differences in Root Architecture, Lignin, and Starch Metabolism and Their Developmental Fates.Front Plant Sci. 2021 Jan 21;11:609923. doi: 10.3389/fpls.2020.609923. eCollection 2020. Front Plant Sci. 2021. PMID: 33552103 Free PMC article.
-
Exogenous gibberellin enhances secondary xylem development and lignification in carrot taproot.Protoplasma. 2017 Mar;254(2):839-848. doi: 10.1007/s00709-016-0995-6. Epub 2016 Jun 22. Protoplasma. 2017. PMID: 27335006
-
Autofluorescence in Plants.Molecules. 2020 May 21;25(10):2393. doi: 10.3390/molecules25102393. Molecules. 2020. PMID: 32455605 Free PMC article. Review.
-
The molecular architecture distinctions between compression, opposite and normal wood of Pinus radiata.Front Plant Sci. 2025 May 13;16:1576928. doi: 10.3389/fpls.2025.1576928. eCollection 2025. Front Plant Sci. 2025. PMID: 40433164 Free PMC article.
-
REPRISAL: mapping lignification dynamics using chemistry, data segmentation, and ratiometric analysis.Plant Physiol. 2022 Feb 4;188(2):816-830. doi: 10.1093/plphys/kiab490. Plant Physiol. 2022. PMID: 34687294 Free PMC article.
References
-
- Abramoff MD, Magelhaes PJ, Ram SJ. (2004) Image Processing with ImageJ. Biophotonics International 11: 36–42
-
- Altaner C, Hapca AI, Knox JP, Jarvis MC. (2007a) Detection of β-1-4-galactan in compression wood of Sitka spruce [Picea sitchensis (Bong.) Carrière] by immunofluorescence. Holzforschung 61: 311–316
-
- Altaner C, Knox JP, Jarvis MC. (2007b) In-situ detection of cell wall polysaccharides in sitka spruce (Picea sitchensis (Bong.) Carrière) wood tissue. BioResources 2: 284–295
-
- Altaner CM, Tokareva EN, Jarvis MC, Harris PJ. (2010) Distribution of (1->4)-β-galactans, arabinogalactan proteins, xylans and (1->3)-β-glucans in tracheid cell walls of softwoods. Tree Physiol 30: 782–793 - PubMed
-
- Arend M. (2008) Immunolocalization of (1,4)-β-galactan in tension wood fibers of poplar. Tree Physiol 28: 1263–1267 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

