Extensive determination of glycan heterogeneity reveals an unusual abundance of high mannose glycans in enriched plasma membranes of human embryonic stem cells
- PMID: 22147732
- PMCID: PMC3322563
- DOI: 10.1074/mcp.M111.010660
Extensive determination of glycan heterogeneity reveals an unusual abundance of high mannose glycans in enriched plasma membranes of human embryonic stem cells
Abstract
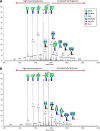
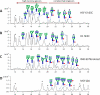
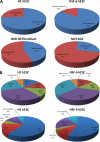
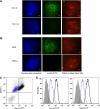
Most cell membrane proteins are known or predicted to be glycosylated in eukaryotic organisms, where surface glycans are essential in many biological processes including cell development and differentiation. Nonetheless, the glycosylation on cell membranes remains not well characterized because of the lack of sensitive analytical methods. This study introduces a technique for the rapid profiling and quantitation of N- and O-glycans on cell membranes using membrane enrichment and nanoflow liquid chromatography/mass spectrometry of native structures. Using this new method, the glycome analysis of cell membranes isolated from human embryonic stem cells and somatic cell lines was performed. Human embryonic stem cells were found to have high levels of high mannose glycans, which contrasts with IMR-90 fibroblasts and a human normal breast cell line, where complex glycans are by far the most abundant and high mannose glycans are minor components. O-Glycosylation affects relatively minor components of cell surfaces. To verify the quantitation and localization of glycans on the human embryonic stem cell membranes, flow cytometry and immunocytochemistry were performed. Proteomics analyses were also performed and confirmed enrichment of plasma membrane proteins with some contamination from endoplasmic reticulum and other membranes. These findings suggest that high mannose glycans are the major component of cell surface glycosylation with even terminal glucoses. High mannose glycans are not commonly presented on the surfaces of mammalian cells or in serum yet may play important roles in stem cell biology. The results also mean that distinguishing stem cells from other mammalian cells may be facilitated by the major difference in the glycosylation of the cell membrane. The deep structural analysis enabled by this new method will enable future mechanistic studies on the biological significance of high mannose glycans on stem cell membranes and provide a general tool to examine cell surface glycosylation.
Figures

Similar articles
-
Deep Structural Analysis and Quantitation of O-Linked Glycans on Cell Membrane Reveal High Abundances and Distinct Glycomic Profiles Associated with Cell Type and Stages of Differentiation.Anal Chem. 2020 Mar 3;92(5):3758-3768. doi: 10.1021/acs.analchem.9b05103. Epub 2020 Feb 20. Anal Chem. 2020. PMID: 32039582 Free PMC article.
-
Regulation of glycan structures in murine embryonic stem cells: combined transcript profiling of glycan-related genes and glycan structural analysis.J Biol Chem. 2012 Nov 2;287(45):37835-56. doi: 10.1074/jbc.M112.405233. Epub 2012 Sep 17. J Biol Chem. 2012. PMID: 22988249 Free PMC article.
-
Region-Specific Cell Membrane N-Glycome of Functional Mouse Brain Areas Revealed by nanoLC-MS Analysis.Mol Cell Proteomics. 2021;20:100130. doi: 10.1016/j.mcpro.2021.100130. Epub 2021 Aug 4. Mol Cell Proteomics. 2021. PMID: 34358619 Free PMC article.
-
O-mannosyl glycans in mammals.Biochim Biophys Acta. 1999 Dec 6;1473(1):237-46. doi: 10.1016/s0304-4165(99)00182-8. Biochim Biophys Acta. 1999. PMID: 10580142 Review.
-
Mass Spectrometry-Based Chemical and Enzymatic Methods for Global Analysis of Protein Glycosylation.Acc Chem Res. 2018 Aug 21;51(8):1796-1806. doi: 10.1021/acs.accounts.8b00200. Epub 2018 Jul 16. Acc Chem Res. 2018. PMID: 30011186 Free PMC article. Review.
Cited by
-
Differentiation of cancer cell origin and molecular subtype by plasma membrane N-glycan profiling.J Proteome Res. 2014 Feb 7;13(2):961-8. doi: 10.1021/pr400987f. Epub 2014 Jan 8. J Proteome Res. 2014. PMID: 24303873 Free PMC article.
-
Deep Structural Analysis and Quantitation of O-Linked Glycans on Cell Membrane Reveal High Abundances and Distinct Glycomic Profiles Associated with Cell Type and Stages of Differentiation.Anal Chem. 2020 Mar 3;92(5):3758-3768. doi: 10.1021/acs.analchem.9b05103. Epub 2020 Feb 20. Anal Chem. 2020. PMID: 32039582 Free PMC article.
-
The effects of immortalization on the N-glycome and proteome of CDK4-transformed lung cancer cells.Glycobiology. 2024 Apr 24;34(6):cwae030. doi: 10.1093/glycob/cwae030. Glycobiology. 2024. PMID: 38579012 Free PMC article.
-
Targeted Analysis of Permethylated N-Glycans Using MRM/PRM Approaches.Methods Mol Biol. 2024;2762:251-266. doi: 10.1007/978-1-0716-3666-4_15. Methods Mol Biol. 2024. PMID: 38315370 Free PMC article.
-
Isomer-specific LC/MS and LC/MS/MS profiling of the mouse serum N-glycome revealing a number of novel sialylated N-glycans.Anal Chem. 2013 May 7;85(9):4636-43. doi: 10.1021/ac400195h. Epub 2013 Apr 18. Anal Chem. 2013. PMID: 23534819 Free PMC article.
References
-
- Apweiler R., Hermjakob H., Sharon N. (1999) On the frequency of protein glycosylation, as deduced from analysis of the SWISS-PROT database. Biochim. Biophys. Acta 1473, 4–8 - PubMed
-
- Ohtsubo K., Marth J. D. (2006) Glycosylation in cellular mechanisms of health and disease. Cell 126, 855–867 - PubMed
-
- Paulson J. C., Blixt O., Collins B. E. (2006) Sweet spots in functional glycomics. Nat. Chem. Biol. 2, 238–248 - PubMed
-
- Dube D. H., Bertozzi C. R. (2005) Glycans in cancer and inflammation: Potential for therapeutics and diagnostics. Nat. Rev. Drug Discov. 4, 477–488 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

