Systemic dissemination of Clostridium difficile toxins A and B is associated with severe, fatal disease in animal models
- PMID: 22147798
- PMCID: PMC3256947
- DOI: 10.1093/infdis/jir748
Systemic dissemination of Clostridium difficile toxins A and B is associated with severe, fatal disease in animal models
Abstract
Background: Clostridium difficile infection (CDI) can cause a wide range of disease, from mild diarrhea to fulminant systemic disease. The incidence of systemic CDI with fatal consequence has increased rapidly in recent years.
Methods: Using an ultrasensitive cytotoxicity assay, we measured C. difficile toxin A (TcdA) and C. difficile toxin B (TcdB) in sera and body fluids of piglets and mice exposed to C. difficile to investigate the relationship between the presence of toxins in body fluids and systemic manifestations of CDI.
Results: We found that both TcdA and TcdB disseminate systemically, with toxins present in the sera and body fluids of infected animals, and toxemia is significantly correlated with the development of systemic CDI. The systemic administration of neutralizing antibodies against both toxins blocked the development of systemic disease in mice. We measured cytokine concentrations in the sera of mice and piglets with systemic and nonsystemic CDI and found that proinflammatory mediators were considerably elevated in animals with systemic CDI.
Conclusion: Our study demonstrates the existence of a strong correlation between toxemia and the occurrence of systemic disease, supporting the hypothesis that systemic CDI is most likely due to the toxicity of TcdA and TcdB and the induction of proinflammatory cytokines by the toxins.
Figures




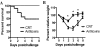
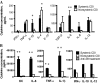
Comment in
-
Clostridium difficile toxins and severe C. difficile infection.J Infect Dis. 2012 Feb 1;205(3):353-4. doi: 10.1093/infdis/jir752. Epub 2011 Dec 5. J Infect Dis. 2012. PMID: 22147800 No abstract available.
Similar articles
-
Defining the Roles of TcdA and TcdB in Localized Gastrointestinal Disease, Systemic Organ Damage, and the Host Response during Clostridium difficile Infections.mBio. 2015 Jun 2;6(3):e00551. doi: 10.1128/mBio.00551-15. mBio. 2015. PMID: 26037121 Free PMC article.
-
Intrarectal instillation of Clostridium difficile toxin A triggers colonic inflammation and tissue damage: development of a novel and efficient mouse model of Clostridium difficile toxin exposure.Infect Immun. 2012 Dec;80(12):4474-84. doi: 10.1128/IAI.00933-12. Epub 2012 Oct 8. Infect Immun. 2012. PMID: 23045481 Free PMC article.
-
Novel Clostridium difficile Anti-Toxin (TcdA and TcdB) Humanized Monoclonal Antibodies Demonstrate In Vitro Neutralization across a Broad Spectrum of Clinical Strains and In Vivo Potency in a Hamster Spore Challenge Model.PLoS One. 2016 Jun 23;11(6):e0157970. doi: 10.1371/journal.pone.0157970. eCollection 2016. PLoS One. 2016. PMID: 27336843 Free PMC article.
-
The enterotoxicity of Clostridium difficile toxins.Toxins (Basel). 2010 Jul;2(7):1848-80. doi: 10.3390/toxins2071848. Epub 2010 Jul 14. Toxins (Basel). 2010. PMID: 22069662 Free PMC article. Review.
-
Clostridium difficile Toxins A and B: Insights into Pathogenic Properties and Extraintestinal Effects.Toxins (Basel). 2016 May 3;8(5):134. doi: 10.3390/toxins8050134. Toxins (Basel). 2016. PMID: 27153087 Free PMC article. Review.
Cited by
-
High Serum Levels of Toxin A Correlate with Disease Severity in Patients with Clostridioides difficile Infection.Antibiotics (Basel). 2021 Sep 10;10(9):1093. doi: 10.3390/antibiotics10091093. Antibiotics (Basel). 2021. PMID: 34572675 Free PMC article.
-
Proinflammatory Cytokines: Possible Accomplices for the Systemic Effects of Clostridioides difficile Toxin B.J Inflamm Res. 2021 Jan 11;14:57-62. doi: 10.2147/JIR.S287096. eCollection 2021. J Inflamm Res. 2021. PMID: 33469335 Free PMC article.
-
A novel multivalent, single-domain antibody targeting TcdA and TcdB prevents fulminant Clostridium difficile infection in mice.J Infect Dis. 2014 Sep 15;210(6):964-72. doi: 10.1093/infdis/jiu196. Epub 2014 Mar 27. J Infect Dis. 2014. PMID: 24683195 Free PMC article.
-
MBX-500 is effective for treatment of Clostridium difficile infection in gnotobiotic piglets.Antimicrob Agents Chemother. 2013 Aug;57(8):4039-41. doi: 10.1128/AAC.00304-13. Epub 2013 May 20. Antimicrob Agents Chemother. 2013. PMID: 23689716 Free PMC article.
-
Newly identified bacteriolytic enzymes that target a wide range of clinical isolates of Clostridium difficile.Biotechnol Bioeng. 2016 Dec;113(12):2568-2576. doi: 10.1002/bit.26029. Epub 2016 Jun 20. Biotechnol Bioeng. 2016. PMID: 27260850 Free PMC article.
References
-
- Elliott B, Chang BJ, Golledge CL, Riley TV. Clostridium difficile-associated diarrhoea. Intern Med J. 2007;37:561–8. - PubMed
-
- Blossom DB, McDonald LC. The challenges posed by reemerging Clostridium difficile infection. Clin Infect Dis. 2007;45:222–7. - PubMed
-
- Kuehne SA, Cartman ST, Heap JT, Kelly ML, Cockayne A, Minton NP. The role of toxin A and toxin B in Clostridium difficile infection. Nature. 2010;467:711–13. - PubMed
-
- Jafri SF, Marshall JB. Ascites associated with antibiotic-associated pseudomembranous colitis. South Med J. 1996;89:1014–7. - PubMed
-
- Tsourous GI, Raftopoulos LG, Kafe EE, Manoleris EK, Makaritsis KP, Pinis SG. A case of pseudomembranous colitis presenting with massive ascites. Eur J Intern Med. 2007;18:328–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

