Vagal control of satiety and hormonal regulation of appetite
- PMID: 22148102
- PMCID: PMC3228973
- DOI: 10.5056/jnm.2011.17.4.338
Vagal control of satiety and hormonal regulation of appetite
Abstract
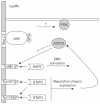
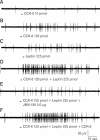
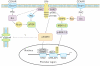
The paradigm for the control of feeding behavior has changed significantly. In this review, we present evidence that the separation of function in which cholecystokinin (CCK) controls short-term food intake and leptin regulate long-term eating behavior and body weight become less clear. In addition to the hypothalamus, the vagus nerve is critically involved in the control of feeding by transmitting signals arising from the upper gut to the nucleus of the solitary tract. Among the peripheral mediators, CCK is the key peptide involved in generating the satiety signal via the vagus. Leptin receptors have also been identified in the vagus nerve. Studies in the rodents clearly indicate that leptin and CCK interact synergistically to induce short-term inhibition of food intake and long-term reduction of body weight. The synergistic interaction between vagal CCK-A receptor and leptin is mediated by the phosphorylation of signal transducer and activator of transcription3 (STAT3), which in turn, activates closure of K(+) channels, leading to membrane depolarization and neuronal firing. This involves the interaction between CCK/SRC/phosphoinositide 3-kinase cascades and leptin/Janus kinase-2/phosphoinositide 3-kinase/STAT3 signaling pathways. It is conceivable that malfunctioning of these signaling molecules may result in eating disorders.
Keywords: Cholecystokinin; Leptin; Nodose ganglion; Signal transduction.
Conflict of interest statement
Conflicts of interest: None.
Figures

Similar articles
-
Synergistic interaction between leptin and cholecystokinin in the rat nodose ganglia is mediated by PI3K and STAT3 signaling pathways: implications for leptin as a regulator of short term satiety.J Biol Chem. 2011 Apr 1;286(13):11707-15. doi: 10.1074/jbc.M110.198945. Epub 2011 Jan 26. J Biol Chem. 2011. PMID: 21270124 Free PMC article.
-
Cocaine- and amphetamine-regulated transcript is the neurotransmitter regulating the action of cholecystokinin and leptin on short-term satiety in rats.Am J Physiol Gastrointest Liver Physiol. 2012 Nov 1;303(9):G1042-51. doi: 10.1152/ajpgi.00231.2012. Epub 2012 Aug 30. Am J Physiol Gastrointest Liver Physiol. 2012. Retraction in: Am J Physiol Gastrointest Liver Physiol. 2023 Feb 1;324(2):G157. doi: 10.1152/ajpgi.00231.2012_RET. PMID: 22936273 Free PMC article. Retracted.
-
KATP channels in the nodose ganglia mediate the orexigenic actions of ghrelin.J Physiol. 2015 Sep 1;593(17):3973-89. doi: 10.1113/JP270788. J Physiol. 2015. Retraction in: J Physiol. 2023 May;601(10):2049. doi: 10.1113/JP284696. PMID: 26174421 Free PMC article. Retracted.
-
Afferent signals regulating food intake.Proc Nutr Soc. 2000 Aug;59(3):373-84. doi: 10.1017/s0029665100000422. Proc Nutr Soc. 2000. PMID: 10997653 Review.
-
Modulation of vagal afferent excitation and reduction of food intake by leptin and cholecystokinin.Physiol Behav. 2006 Nov 30;89(4):477-85. doi: 10.1016/j.physbeh.2006.06.017. Epub 2006 Jul 26. Physiol Behav. 2006. PMID: 16872644 Review.
Cited by
-
Vagus Nerve as Modulator of the Brain-Gut Axis in Psychiatric and Inflammatory Disorders.Front Psychiatry. 2018 Mar 13;9:44. doi: 10.3389/fpsyt.2018.00044. eCollection 2018. Front Psychiatry. 2018. PMID: 29593576 Free PMC article. Review.
-
Loss of Vagal Sensitivity to Cholecystokinin in Rats Born with Intrauterine Growth Retardation and Consequence on Food Intake.Front Endocrinol (Lausanne). 2017 Apr 10;8:65. doi: 10.3389/fendo.2017.00065. eCollection 2017. Front Endocrinol (Lausanne). 2017. PMID: 28443064 Free PMC article.
-
Weight Regain after Metabolic Surgery: Beyond the Surgical Failure.J Clin Med. 2024 Feb 18;13(4):1143. doi: 10.3390/jcm13041143. J Clin Med. 2024. PMID: 38398456 Free PMC article. Review.
-
Decreased expression of the satiety signal receptor CCKAR is responsible for increased growth and body weight during the domestication of chickens.Am J Physiol Endocrinol Metab. 2013 May 1;304(9):E909-21. doi: 10.1152/ajpendo.00580.2012. Epub 2013 Feb 26. Am J Physiol Endocrinol Metab. 2013. PMID: 23443924 Free PMC article.
-
Vagal neurocircuitry and its influence on gastric motility.Nat Rev Gastroenterol Hepatol. 2016 Jul;13(7):389-401. doi: 10.1038/nrgastro.2016.76. Epub 2016 May 25. Nat Rev Gastroenterol Hepatol. 2016. PMID: 27251213 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

