A single sublingual dose of an adenovirus-based vaccine protects against lethal Ebola challenge in mice and guinea pigs
- PMID: 22149096
- PMCID: PMC3358355
- DOI: 10.1021/mp200392g
A single sublingual dose of an adenovirus-based vaccine protects against lethal Ebola challenge in mice and guinea pigs
Abstract
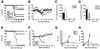
Sublingual (SL) delivery, a noninvasive immunization method that bypasses the intestinal tract for direct entry into the circulation, was evaluated with an adenovirus (Ad5)-based vaccine for Ebola. Mice and guinea pigs were immunized via the intramuscular (IM), nasal (IN), oral (PO) and SL routes. SL immunization elicited strong transgene expression in and attracted CD11c(+) antigen presenting cells to the mucosa. A SL dose of 1 × 10⁸ infectious particles induced Ebola Zaire glycoprotein (ZGP)-specific IFN-γ⁺ T cells in spleen, bronchoalveolar lavage, mesenteric lymph nodes and submandibular lymph nodes (SMLN) of naive mice in a manner similar to the same dose given IN. Ex vivo CFSE and in vivo cytotoxic T lymphocyte (CTL) assays confirmed that SL immunization elicits a notable population of effector memory CD8+ T cells and strong CTL responses in spleen and SMLN. SL immunization induced significant ZGP-specific Th1 and Th2 type responses unaffected by pre-existing immunity (PEI) that protected mice and guinea pigs from lethal challenge. SL delivery protected more mice with PEI to Ad5 than IM injection. SL immunization also reduced systemic anti-Ad5 T and B cell responses in naive mice and those with PEI, suggesting that secondary immunizations could be highly effective for both populations.
Figures





References
-
- Richardson JS, Dekker JD, Croyle MA, Kobinger GP. Recent Advances in Ebolavirus Vaccine Development. Hum. Vaccine. 2010;6(6):439–449. - PubMed
-
- Towner JS, Sealy TK, Khristova ML, Albariño CG, Conlan S, Reeder SA, Quan PL, Lipkin WI, Downing R, Tappero JW, Okware S, Lutwama J, Bakamutumaho B, Kayiwa J, Comer JA, Rollin PE, Ksiazek TG, Nichol ST. Newly discovered ebola virus associated with hemorrhagic fever outbreak in Uganda. PLoS Pathog. 2008;4(11):e1000212. - PMC - PubMed
-
- Formenty P, Hatz C, Le Guenno B, Stoll A, Rogenmoser P, Widmer A. Human infection due to Ebola virus, subtype Côte d'Ivoire: clinical and biologic presentation. J. Infect. Dis. 1999;179(Suppl. 1):S48–S53. - PubMed
-
- Barrette RW, Metwally SA, Rowland JM, Xu L, Zaki SR, Nichol ST, Rollin PE, Towner JS, Shieh WJ, Batten B, Sealy TK, Carrillo C, Moran KE, Bracht AJ, Mayr GA, Sirios-Cruz M, Catbagan DP, Lautner EA, Ksiazek TG, White WR, McIntosh MT. Discovery of swine as a host for the Reston ebolavirus. Science. 2009;325(5397):204–206. - PubMed
-
- Hartman AL, Towner JS, Nichol ST. Ebola and Marburg Hemorrhagic Fever. Clin. Lab. Med. 2010;30(1):161–177. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

