Roles of hydrogen sulfide in the pathogenesis of diabetes mellitus and its complications
- PMID: 22149162
- PMCID: PMC4701125
- DOI: 10.1089/ars.2011.4451
Roles of hydrogen sulfide in the pathogenesis of diabetes mellitus and its complications
Abstract
Significance: Diabetes and its complications represent a major socioeconomic problem.
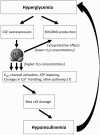
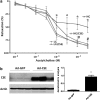
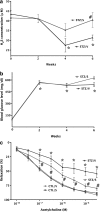
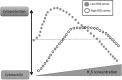
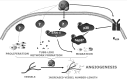
Recent advances: Changes in the balance of hydrogen sulfide (H(2)S) play an important role in the pathogenesis of β-cell dysfunction that occurs in response to type 1 and type 2 diabetes. In addition, changes in H(2)S homeostasis also play a role in the pathogenesis of endothelial injury, which develop on the basis of chronically or intermittently elevated circulating glucose levels in diabetes.
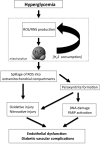
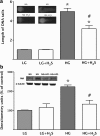
Critical issues: In the first part of this review, experimental evidence is summarized implicating H(2)S overproduction as a causative factor in the pathogenesis of β-cell death in diabetes. In the second part of our review, experimental evidence is presented supporting the role of H(2)S deficiency (as a result of increased H(2)S consumption by hyperglycemic cells) in the pathogenesis of diabetic endothelial dysfunction, diabetic nephropathy, and cardiomyopathy.
Future directions: In the final section of the review, future research directions and potential experimental therapeutic approaches around the pharmacological modulation of H(2)S homeostasis in diabetes are discussed.
Figures

References
-
- Ali MY. Whiteman M. Low CM. Moore PK. Hydrogen sulphide reduces insulin secretion from HIT-T15 cells by a KATP channel-dependent pathway. J Endocrinol. 2007;195:105–112. - PubMed
-
- Baluchnejadmojarad T. Roghani M. Endothelium-dependent and -independent effect of aqueous extract of garlic on vascular reactivity on diabetic rats. Fitoterapia. 2003;74:630–637. - PubMed
-
- Baskar R. Li L. Moore PK. Hydrogen sulfide-induces DNA damage and changes in apoptotic gene expression in human lung fibroblast cells. FASEB J. 2007;21:247–255. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

