Functional impairment of Tax-specific but not cytomegalovirus-specific CD8+ T lymphocytes in a minor population of asymptomatic human T-cell leukemia virus type 1-carriers
- PMID: 22151736
- PMCID: PMC3261825
- DOI: 10.1186/1742-4690-8-100
Functional impairment of Tax-specific but not cytomegalovirus-specific CD8+ T lymphocytes in a minor population of asymptomatic human T-cell leukemia virus type 1-carriers
Abstract
Background: Human T-cell leukemia virus type 1 (HTLV-1) causes adult T-cell leukemia (ATL) and HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP) in a small percentage of infected individuals. ATL is often associated with general immune suppression and an impaired HTLV-1-specific T-cell response, an important host defense system. We previously found that a small fraction of asymptomatic HTLV-1-carriers (AC) already showed impaired T-cell responses against the major target antigen, Tax. However, it is unclear whether the impaired HTLV-1 Tax-specific T-cell response in these individuals is an HTLV-1-specific phenomenon, or merely reflects general immune suppression. In this study, in order to characterize the impaired HTLV-1-specific T-cell response, we investigated the function of Tax-specific CD8+ T-cells in various clinical status of HTLV-1 infection.
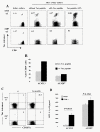
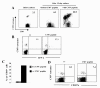
Results: By using tetramers consisting of HLA-A*0201, -A*2402, or -A*1101, and corresponding Tax epitope peptides, we detected Tax-specific CD8+ T-cells in the peripheral blood from 87.0% of ACs (n = 20/23) and 100% of HAM/TSP patients (n = 18/18) tested. We also detected Tax-specific CD8+ T-cells in 38.1% of chronic type ATL (cATL) patients (n = 8/21), although its frequencies in peripheral blood CD8+ T cells were significantly lower than those of ACs or HAM/TSP patients. Tax-specific CD8+ T-cells detected in HAM/TSP patients proliferated well in culture and produced IFN-γ when stimulated with Tax peptides. However, such functions were severely impaired in the Tax-specific CD8+ T-cells detected in cATL patients. In ACs, the responses of Tax-specific CD8+ T-cells were retained in most cases. However, we found one AC sample whose Tax-specific CD8+ T-cells hardly produced IFN-γ, and failed to proliferate and express activation (CD69) and degranulation (CD107a) markers in response to Tax peptide. Importantly, the same AC sample contained cytomegalovirus (CMV) pp65-specific CD8+ T-cells that possessed functions upon CMV pp65 peptide stimulation. We further examined additional samples of two smoldering type ATL patients and found that they also showed dysfunctions of Tax-specific but not CMV-specific CD8+ T-cells.
Conclusions: These findings indicated that Tax-specific CD8+ T-cells were scarce and dysfunctional not only in ATL patients but also in a limited AC population, and that the dysfunction was selective for HTLV-1-specifc CD8+ T-cells in early stages.
Figures




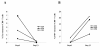
Similar articles
-
Impact of host immunity on HTLV-1 pathogenesis: potential of Tax-targeted immunotherapy against ATL.Retrovirology. 2019 Aug 22;16(1):23. doi: 10.1186/s12977-019-0484-z. Retrovirology. 2019. PMID: 31438973 Free PMC article. Review.
-
Lack of recall response to Tax in ATL and HAM/TSP patients but not in asymptomatic carriers of human T-cell leukemia virus type 1.J Clin Immunol. 2013 Oct;33(7):1223-39. doi: 10.1007/s10875-013-9918-x. Epub 2013 Jul 26. J Clin Immunol. 2013. PMID: 23888327 Free PMC article.
-
IFN-gamma production in response to Tax 161-233, and frequency of CD4+ Foxp3+ and Lin HLA-DRhigh CD123+ cells, discriminate HAM/TSP patients from asymptomatic HTLV-1-carriers in a Peruvian population.Immunology. 2009 Sep;128(1 Suppl):e777-86. doi: 10.1111/j.1365-2567.2009.03082.x. Epub 2009 Feb 17. Immunology. 2009. PMID: 19740339 Free PMC article.
-
Demonstration of human T lymphotropic virus type I (HTLV-I) tax-specific CD8+ lymphocytes directly in peripheral blood of HTLV-I-associated myelopathy/tropical spastic paraparesis patients by intracellular cytokine detection.J Immunol. 1998 Jul 1;161(1):482-8. J Immunol. 1998. PMID: 9647259
-
Human T-lymphotropic virus type 1 (HTLV-1) and cellular immune response in HTLV-1-associated myelopathy/tropical spastic paraparesis.J Neurovirol. 2020 Oct;26(5):652-663. doi: 10.1007/s13365-020-00881-w. Epub 2020 Jul 23. J Neurovirol. 2020. PMID: 32705480 Free PMC article. Review.
Cited by
-
Maintenance of long remission in adult T-cell leukemia by Tax-targeted vaccine: A hope for disease-preventive therapy.Cancer Sci. 2019 Mar;110(3):849-857. doi: 10.1111/cas.13948. Epub 2019 Feb 19. Cancer Sci. 2019. PMID: 30666755 Free PMC article. Review.
-
Long-term persistence of limited HTLV-I Tax-specific cytotoxic T cell clones in a patient with adult T cell leukemia/lymphoma after allogeneic stem cell transplantation.J Clin Immunol. 2012 Dec;32(6):1340-52. doi: 10.1007/s10875-012-9729-5. Epub 2012 Jul 5. J Clin Immunol. 2012. PMID: 22763862
-
Diversity matters in scientific publishing.Retrovirology. 2012 Dec 19;9:109. doi: 10.1186/1742-4690-9-109. Retrovirology. 2012. PMID: 23253815 Free PMC article.
-
HTLV-1, the Other Pathogenic Yet Neglected Human Retrovirus: From Transmission to Therapeutic Treatment.Viruses. 2017 Dec 21;10(1):1. doi: 10.3390/v10010001. Viruses. 2017. PMID: 29267225 Free PMC article. Review.
-
Impact of host immunity on HTLV-1 pathogenesis: potential of Tax-targeted immunotherapy against ATL.Retrovirology. 2019 Aug 22;16(1):23. doi: 10.1186/s12977-019-0484-z. Retrovirology. 2019. PMID: 31438973 Free PMC article. Review.
References
-
- de The G, Bomford R. An HTLV-I vaccine: why, how, for whom? AIDS Res Hum Retroviruses. 1993;9:381–386. - PubMed
-
- Arisawa K, Soda M, Endo S, Kurokawa K, Katamine S, Shimokawa I, Koba T, Takahashi T, Saito H, Doi H, Shirahama S. Evaluation of adult T-cell leukemia/lymphoma incidence and its impact on non-Hodgkin lymphoma incidence in southwestern Japan. Int J Cancer. 2000;85:319–324. - PubMed
-
- Gessain A, Barin F, Vernant JC, Gout O, Maurs L, Calender A, de The G. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet. 1985;2:407–410. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

