Influence of cAMP and protein kinase A on neurite length from spiral ganglion neurons
- PMID: 22154930
- PMCID: PMC3277666
- DOI: 10.1016/j.heares.2011.11.010
Influence of cAMP and protein kinase A on neurite length from spiral ganglion neurons
Abstract
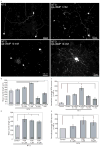
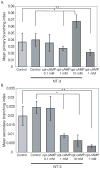
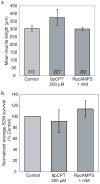
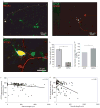
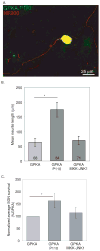
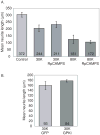
Regrowth of peripheral spiral ganglion neuron (SGN) fibers is a primary objective in efforts to improve cochlear implant outcomes and to potentially reinnervate regenerated hair cells. Cyclic adenosine monophosphate (cAMP) regulates neurite growth and guidance via activation of protein kinase A (PKA) and Exchange Protein directly Activated by Cylic AMP (Epac). Here we explored the effects of cAMP signaling on SGN neurite length in vitro. We find that the cAMP analog, cpt-cAMP, exerts a biphasic effect on neurite length; increasing length at lower concentrations and reducing length at higher concentrations. This biphasic response occurs in cultures plated on laminin, fibronectin, or tenascin C suggesting that it is not substrate dependent. cpt-cAMP also reduces SGN neurite branching. The Epac-specific agonist, 8-pCPT-2'-O-Me-cAMP, does not alter SGN neurite length. Constitutively active PKA isoforms strongly inhibit SGN neurite length similar to higher levels of cAMP. Chronic membrane depolarization activates PKA in SGNs and also inhibits SGN neurite length. However, inhibition of PKA fails to rescue neurite length in depolarized cultures implying that activation of PKA is not necessary for the inhibition of SGN neurite length by chronic depolarization. Expression of constitutively active phosphatidylinositol 3-kinase, but not c-Jun N-terminal kinase, isoforms partially rescues SGN neurite length in the presence of activated PKA. Taken together, these results suggest that activation of cAMP/PKA represents a potential strategy to enhance SGN fiber elongation following deafness; however such therapies will likely require careful titration so as to promote rather than inhibit nerve fiber regeneration.
Copyright © 2011 Elsevier B.V. All rights reserved.
Figures


References
-
- Aglah C, Gordon T, Posse de Chaves EI. cAMP promotes neurite outgrowth and extension through protein kinase A but independently of Erk activation in cultured rat motoneurons. Neuropharmacology. 2008;55:8–17. - PubMed
-
- Agterberg MJ, Versnel H, de Groot JC, van den Broek M, Klis SF. Chronic electrical stimulation does not prevent spiral ganglion cell degeneration in deafened guinea pigs. Hearing research. 2010;269:169–79. - PubMed
-
- Alam SA, Robinson BK, Huang J, Green SH. Prosurvival and proapoptotic intracellular signaling in rat spiral ganglion neurons in vivo after the loss of hair cells. J Comp Neurol. 2007;503:832–52. - PubMed
-
- Altschuler RA, Cho Y, Ylikoski J, Pirvola U, Magal E, Miller JM. Rescue and regrowth of sensory nerves following deafferentation by neurotrophic factors. Ann N Y Acad Sci. 1999;884:305–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

