Fak depletion in both hematopoietic and nonhematopoietic niche cells leads to hematopoietic stem cell expansion
- PMID: 22155722
- PMCID: PMC3307834
- DOI: 10.1016/j.exphem.2011.11.010
Fak depletion in both hematopoietic and nonhematopoietic niche cells leads to hematopoietic stem cell expansion
Abstract
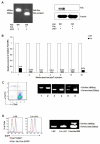
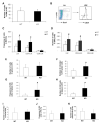
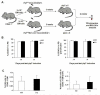
Hematopoietic stem cells (HSCs) reside in complex bone marrow microenvironments, where niche-induced signals regulate hematopoiesis. Focal adhesion kinase (Fak) is a nonreceptor protein tyrosine kinase that plays an essential role in many cell types, where its activation controls adhesion, motility, and survival. Fak expression is relatively increased in HSCs compared to progenitors and mature blood cells. Therefore, we explored its role in HSC homeostasis. We have used the Mx1-Cre-inducible conditional knockout mouse model to investigate the effects of Fak deletion in bone marrow compartments. The total number as well as the fraction of cycling Lin(-)Sca-1(+)c-kit(+) (LSK) cells is increased in Fak(-/-) mice compared to controls, while hematopoietic progenitors and mature blood cells are unaffected. Bone marrow cells from Fak(-/-) mice exhibit enhanced, long-term (i.e., 20-week duration) engraftment in competitive transplantation assays. Intrinsic Fak function was assessed in serial transplantation assays, which showed that HSCs (Lin(-)Sca-1(+)c-kit(+)CD34(-)Flk-2(-) cells) sorted from Fak(-/-) mice have similar self-renewal and engraftment ability on a per-cell basis as wild-type HSCs. When Fak deletion is induced after engraftment of Fak(fl/fl)Mx1-Cre(+) bone marrow cells into wild-type recipient mice, the number of LSKs is unchanged. In conclusion, Fak inactivation does not intrinsically regulate HSC behavior and is not essential for steady-state hematopoiesis. However, widespread Fak inactivation in the hematopoietic system induces an increased and activated HSC pool size, potentially as a result of altered reciprocal interactions between HSCs and their microenvironment.
Copyright © 2012 ISEH - Society for Hematology and Stem Cells. Published by Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Ash1l controls quiescence and self-renewal potential in hematopoietic stem cells.J Clin Invest. 2015 May;125(5):2007-20. doi: 10.1172/JCI78124. Epub 2015 Apr 13. J Clin Invest. 2015. PMID: 25866973 Free PMC article.
-
Phenotypic analysis and isolation of murine hematopoietic stem cells and lineage-committed progenitors.J Vis Exp. 2012 Jul 8;(65):3736. doi: 10.3791/3736. J Vis Exp. 2012. PMID: 22805770 Free PMC article.
-
Impact of interactions of cellular components of the bone marrow microenvironment on hematopoietic stem and progenitor cell function.Blood. 2010 Apr 22;115(16):3239-48. doi: 10.1182/blood-2009-09-246173. Epub 2010 Feb 12. Blood. 2010. PMID: 20154218 Free PMC article.
-
Role of the microenvironment of the embryonic aorta-gonad-mesonephros region in hematopoiesis.Ann N Y Acad Sci. 2001 Jun;938:109-16. doi: 10.1111/j.1749-6632.2001.tb03579.x. Ann N Y Acad Sci. 2001. PMID: 11458497 Review.
-
Hematopoietic Stem Cells, Their Niche, and the Concept of Co-Culture Systems: A Critical Review.J Stem Cells. 2015;10(1):13-31. J Stem Cells. 2015. PMID: 26665935 Review.
Cited by
-
Maintenance of hematopoietic stem cell dormancy: yet another role for the macrophage.Stem Cell Investig. 2016 Sep 19;3:46. doi: 10.21037/sci.2016.08.10. eCollection 2016. Stem Cell Investig. 2016. PMID: 27777935 Free PMC article. No abstract available.
-
Mechanobiology of bone marrow stem cells: from myosin-II forces to compliance of matrix and nucleus in cell forms and fates.Differentiation. 2013 Oct;86(3):77-86. doi: 10.1016/j.diff.2013.05.001. Epub 2013 Jun 19. Differentiation. 2013. PMID: 23790394 Free PMC article. Review.
-
Hypoxic niche-mediated regeneration of hematopoiesis in the engraftment window is dominantly affected by oxygen tension in the milieu.Stem Cells Dev. 2015 Oct 15;24(20):2423-36. doi: 10.1089/scd.2015.0112. Epub 2015 Jul 28. Stem Cells Dev. 2015. PMID: 26107807 Free PMC article.
-
Modeling the Pro-inflammatory Tumor Microenvironment in Acute Lymphoblastic Leukemia Predicts a Breakdown of Hematopoietic-Mesenchymal Communication Networks.Front Physiol. 2016 Aug 19;7:349. doi: 10.3389/fphys.2016.00349. eCollection 2016. Front Physiol. 2016. PMID: 27594840 Free PMC article.
-
Adhesion in the stem cell niche: biological roles and regulation.Development. 2013 Jan 15;140(2):255-65. doi: 10.1242/dev.083139. Development. 2013. PMID: 23250203 Free PMC article. Review.
References
-
- Weissman IL. Stem cells: units of development, units of regeneration, and units in evolution. Cell. 2000;100:157–168. - PubMed
-
- Hardy RR, Hayakawa K. B cell development pathways. Annu Rev Immunol. 2001;19:595–621. - PubMed
-
- Yang L, Bryder D, Adolfsson J, et al. Identification of Lin(−)Sca1(+)kit(+)CD34(+)Flt3− short-term hematopoietic stem cells capable of rapidly reconstituting and rescuing myeloablated transplant recipients. Blood. 2005;105:2717–2723. - PubMed
-
- Adolfsson J, Mansson R, Buza-Vidas N, et al. Identification of Flt3+ lympho-myeloid stem cells lacking erythro-megakaryocytic potential a revised road map for adult blood lineage commitment. Cell. 2005;121:295–306. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

