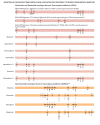
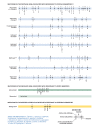
2011 update of the drug resistance mutations in HIV-1
- PMID: 22156218
- PMCID: PMC6148877
2011 update of the drug resistance mutations in HIV-1
Abstract
This November 2011 edition of the IAS-USA drug resistance mutations list updates the figures last published in December 2010 (Johnson VA et al, Top HIV Med, 2010;18:156-163).
Conflict of interest statement
Financial Disclosures: The authors (listed alphabetically) disclose the following affiliations with commercial organizations that may have interests related to the content of this article (previous 12 months): Dr Calvez has served on advisory boards for Abbott Laboratories, Bristol-Myers Squibb, Gilead Sciences, Inc, GlaxoSmithKline, Janssen Pharmaceutica, Pfizer, Inc, Roche, and ViiV Healthcare. Dr Günthard has served as a consultant or medical advisor for Abbott Laboratories, Boehringer Ingelheim GmbH, Bristol-Myers Squibb, F. Hoffmann-La Roche Ltd, Gilead Sciences, Inc, GlaxoSmithKline, Merck Sharp & Dohme Corp, Novartis AG, and Tibotec Pharmaceuticals; has received unrestricted research grants from Abbott Laboratories, Bristol-Myers Squibb, F. Hoffmann-La Roche Ltd, Gilead Sciences, Inc, GlaxoSmithKline, Merck Sharp & Dohme Corp, Tibotec Pharmaceuticals, and ViiV Healthcare; and has received travel grants from Abbott Laboratories, Bristol-Myers Squibb, F. Hoffmann-La Roche Ltd, Gilead Sciences, Inc, GlaxoSmithKline, Merck Sharp & Dohme Corp, Tibotec Pharmaceuticals, and ViiV Healthcare. Dr Johnson has received research support from Abbott Molecular, Roche Molecular Diagnostics, and Siemens Healthcare Diagnostics Inc. Dr Paredes has served as a consultant or medical advisor for Roche Diagnostics and ViiV Healthcare, and has received research grants awarded to IrsiCaixa and Lluita Contra la SIDA Foundations from Merck Sharp & Dohme Corp and ViiV Healthcare. Dr Pillay has no relevant financial affiliations to disclose. Dr Richman has been a consultant to Biota, Bristol-Myers Squibb, CHIMERx, Gen-Probe Inc, Gilead Sciences, Inc, Idenix Pharmaceuticals, Inc, Johnson & Johnson, Merck & Co, Inc, and Monogram Biosciences, Inc; received research grants or contracts from Merck & Co, Inc; and has been a stock options holder of CHIMERx and Idenix Pharmaceuticals, Inc. Dr Shafer has served as a consultant or medical advisor for Celera and Siemens Healthcare and has received grants from F. Hoffmann-La Roche Ltd and Gilead Sciences, Inc. Dr Wensing has served as a consultant or medical advisor for Bristol-Myers Squibb, Gilead Sciences, Inc, Merck & Co, Inc, and ViiV Healthcare; has received grants from Merck & Co, Inc, and ViiV Healthcare; and has received travel, accommodations, or meeting expenses from Bristol-Myers Squibb, Gilead Sciences, Inc, and Merck & Co, Inc. The IAS–USA has received grants in the past year for selected continuing medical education activities that are pooled (ie, no single company supports any particular effort) from Abbott Laboratories, Bristol-Myers Squibb, Gilead Sciences, Inc, Merck & Co, Inc, Tibotec Therapeutics, Vertex Pharmaceuticals, and ViiV Healthcare.
References
-
- Clark SA, Shulman NS, Bosch RJ, Mellors JW. Reverse transcriptase mutations 118I, 208Y, and 215Y cause HIV-1 hypersusceptibility to non-nucleoside reverse transcriptase inhibitors. AIDS. 2006;20:981-984. - PubMed
-
- Picchio G, Vingerhoets J, Parkin N, Azijn H, de Bethune MP. Nucleoside-associated mutations cause hypersusceptibility to etravirine. [Abstract 23.] Antivir Ther. 2008;13(Suppl 3):A25.
-
- Shulman NS, Bosch RJ, Mellors JW, Albrecht MA, Katzenstein DA. Genetic correlates of efavirenz hypersusceptibility. AIDS. 2004;18:1781-1785. - PubMed
-
- Demeter LM, DeGruttola V, Lustgarten S, et al. Association of efavirenz hypersus-ceptibility with virologic response in ACTG 368, a randomized trial of abacavir (ABC) in combination with efavirenz (EFV) and indinavir (IDV) in HIV-infected subjects with prior nucleoside analog experience. HIV Clin TriSals. 2008;9:11-25. - PMC - PubMed
-
- Haubrich RH, Kemper CA, Hellmann NS, et al. The clinical relevance of nonnucleoside reverse transcriptase inhibitor hypersusceptibility: a prospective cohort analysis. AIDS. 2002;16:F33-F40. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical