Natural killer cells regulate murine cytomegalovirus-induced sialadenitis and salivary gland disease
- PMID: 22156514
- PMCID: PMC3302420
- DOI: 10.1128/JVI.06898-11
Natural killer cells regulate murine cytomegalovirus-induced sialadenitis and salivary gland disease
Abstract
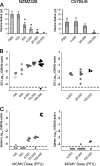
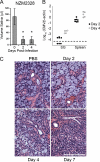
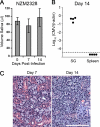
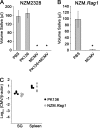
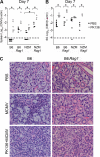
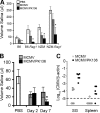
The transmission of herpesviruses depends on viral shedding at mucosal surfaces. The salivary gland represents a major site of persistent viral replication for many viruses, including cytomegalovirus. We established a mouse model of salivary gland dysfunction after acute viral infection and investigated the cellular requirements for the loss of secretion. Murine cytomegalovirus (MCMV) infection severely impaired saliva secretion independently of salivary gland virus levels. Lymphocytes or circulating monocytes/macrophages were not required for secretory dysfunction. Dysfunction occurred before glandular inflammation, suggesting that a soluble mediator initiated the disruption of acinar cell function. Despite genetic differences in innate resistance to MCMV, NK cells protected the host against acinar atrophy and the loss of secretions under conditions of an exceedingly low virus inoculum. NK cells also modulated the type of glandular inflammation after infection, as they prevented an influx of Siglec-F(+) polymorphonuclear leukocytes (PMNs). Therefore, beyond their recognized role in controlling MCMV replication, NK cells preserve organ integrity and function and regulate the innate inflammatory response within the gland.
Figures
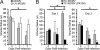

Similar articles
-
Murine Cytomegalovirus MCK-2 Facilitates In Vivo Infection Transfer from Dendritic Cells to Salivary Gland Acinar Cells.J Virol. 2021 Aug 10;95(17):e0069321. doi: 10.1128/JVI.00693-21. Epub 2021 Aug 10. J Virol. 2021. PMID: 34132572 Free PMC article.
-
Natural killer cell dependent within-host competition arises during multiple MCMV infection: consequences for viral transmission and evolution.PLoS Pathog. 2013 Jan;9(1):e1003111. doi: 10.1371/journal.ppat.1003111. Epub 2013 Jan 3. PLoS Pathog. 2013. PMID: 23300458 Free PMC article.
-
EBI3 regulates the NK cell response to mouse cytomegalovirus infection.Proc Natl Acad Sci U S A. 2017 Feb 14;114(7):1625-1630. doi: 10.1073/pnas.1700231114. Epub 2017 Jan 31. Proc Natl Acad Sci U S A. 2017. PMID: 28143936 Free PMC article.
-
The salivary glands as a privileged site of cytomegalovirus immune evasion and persistence.Med Microbiol Immunol. 2008 Jun;197(2):205-13. doi: 10.1007/s00430-008-0077-2. Epub 2008 Feb 8. Med Microbiol Immunol. 2008. PMID: 18259775 Review.
-
Natural killer cell licensing during viral infection.Adv Exp Med Biol. 2011;780:37-44. doi: 10.1007/978-1-4419-5632-3_4. Adv Exp Med Biol. 2011. PMID: 21842363 Review.
Cited by
-
Immune surveillance of cytomegalovirus in tissues.Cell Mol Immunol. 2024 Sep;21(9):959-981. doi: 10.1038/s41423-024-01186-2. Epub 2024 Aug 12. Cell Mol Immunol. 2024. PMID: 39134803 Free PMC article. Review.
-
Friend or Foe? Exploring the Role of Cytomegalovirus (HCMV) Infection in Head and Neck Tumors.Biomedicines. 2024 Apr 15;12(4):872. doi: 10.3390/biomedicines12040872. Biomedicines. 2024. PMID: 38672226 Free PMC article.
-
ILC1s in Tissue Inflammation and Infection.Front Immunol. 2016 Mar 22;7:104. doi: 10.3389/fimmu.2016.00104. eCollection 2016. Front Immunol. 2016. PMID: 27047491 Free PMC article. Review.
-
Phenotype and functions of conventional and non-conventional NK cells.Curr Opin Immunol. 2016 Feb;38:67-74. doi: 10.1016/j.coi.2015.11.007. Epub 2015 Dec 17. Curr Opin Immunol. 2016. PMID: 26706497 Free PMC article. Review.
-
Heightened innate immune state induced by viral vector leads to enhanced response to challenge and prolongs malaria vaccine protection.iScience. 2024 Nov 26;27(12):111468. doi: 10.1016/j.isci.2024.111468. eCollection 2024 Dec 20. iScience. 2024. PMID: 39758993 Free PMC article.
References
-
- Basu D, Williams FM, Ahn CW, Reveille JD. 2006. Changing spectrum of the diffuse infiltrative lymphocytosis syndrome. Arthritis Rheum. 55:466–472 - PubMed
-
- Brown MG, et al. 2001. Vital involvement of a natural killer cell activation receptor in resistance to viral infection. Science 292:934–937 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

