Neuroprotection by leptin in a rat model of permanent cerebral ischemia: effects on STAT3 phosphorylation in discrete cells of the brain
- PMID: 22158477
- PMCID: PMC3252737
- DOI: 10.1038/cddis.2011.125
Neuroprotection by leptin in a rat model of permanent cerebral ischemia: effects on STAT3 phosphorylation in discrete cells of the brain
Abstract
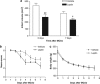
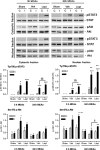
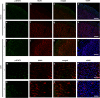
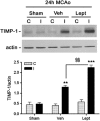
In addition to its effects in the hypothalamus to control body weight, leptin is involved in the regulation of neuronal function, development and survival. Recent findings have highlighted the neuroprotective effects of leptin against ischemic brain injury; however, to date, little is known about the role performed by the signal transducer and activator of transcription (STAT)-3, a major mediator of leptin receptor transduction pathway in the brain, in the beneficial effects of the hormone. Our data demonstrate that systemic acute administration of leptin produces neuroprotection in rats subjected to permanent middle cerebral artery occlusion (MCAo), as revealed by a significant reduction of the brain infarct volume and neurological deficit up to 7 days after the induction of ischemia. By combining a subcellular fractionation approach with immunohistofluorescence, we observe that neuroprotection is associated with a cell type-specific modulation of STAT3 phosphorylation in the ischemic cortex. The early enhancement of nuclear phospho-STAT3 induced by leptin in the astrocytes of the ischemic penumbra may contribute to a beneficial effect of these cells on the evolution of tissue damage. In addition, the elevation of phospho-STAT3 induced by leptin in the neurons after 24 h MCAo is associated with an increased expression of tissue inhibitor of matrix metalloproteinases-1 in the cortex, suggesting its possible involvement to the neuroprotection produced by the adipokine.
Figures



References
-
- Tang BL. Leptin as a neuroprotective agent. Biochem Biophys Res Comm. 2008;368:181–185. - PubMed
-
- Elmquist JK, Bjørbaek C, Ahima RS, Flier JS, Saper CB. Distributions of leptin receptor mRNA isoforms in the rat brain. J Comp Neurol. 1998;395:535–547. - PubMed
-
- Zhang F, Wang S, Signore AP, Chen J. Neuroprotective effects of leptin against ischemic injury induced by oxygen-glucose deprivation and transient cerebral ischemia. Stroke. 2007;38:2329–2336. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

