MT1-MMP regulates the turnover and endocytosis of extracellular matrix fibronectin
- PMID: 22159414
- PMCID: PMC3244985
- DOI: 10.1242/jcs.087858
MT1-MMP regulates the turnover and endocytosis of extracellular matrix fibronectin
Abstract
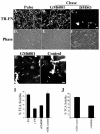
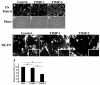
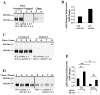
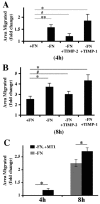
The extracellular matrix (ECM) is dynamically remodeled by cells during development, normal tissue homeostasis and in a variety of disease processes. We previously showed that fibronectin is an important regulator of ECM remodeling. The deposition and/or polymerization of fibronectin into the ECM controls the deposition and stability of other ECM molecules. In addition, agents that inhibit fibronectin polymerization promote the turnover of fibronectin fibrils and enhance ECM fibronectin endocytosis and intracellular degradation. Endocytosis of ECM fibronectin is regulated by β1 integrins, including α5β1 integrin. We have examined the role of extracellular proteases in regulating ECM fibronectin turnover. Our data show that membrane type matrix metalloproteinase 1 (MT1-MMP; also known as MMP14) is a crucial regulator of fibronectin turnover. Cells lacking MT1-MMP show reduced turnover and endocytosis of ECM fibronectin. MT1-MMP regulates ECM fibronectin remodeling by promoting extracellular cleavage of fibronectin and by regulating α5β1-integrin endocytosis. Our data also show that fibronectin polymerization stabilizes fibronectin fibrils and inhibits ECM fibronectin endocytosis by inhibiting α5β1-integrin endocytosis. These data are the first to show that an ECM protein and its modifying enzyme can regulate integrin endocytosis. These data also show that integrin trafficking plays a major role in modulating ECM fibronectin remodeling. The dual dependence of ECM fibronectin turnover on extracellular proteolysis and endocytosis highlights the complex regulatory mechanisms that control ECM remodeling to ensure maintenance of proper tissue function.
Figures






Similar articles
-
Collagen I matrix turnover is regulated by fibronectin polymerization.Am J Physiol Cell Physiol. 2010 May;298(5):C1265-75. doi: 10.1152/ajpcell.00341.2009. Epub 2010 Jan 27. Am J Physiol Cell Physiol. 2010. PMID: 20107040 Free PMC article.
-
Caveolin-1-dependent beta1 integrin endocytosis is a critical regulator of fibronectin turnover.J Cell Sci. 2008 Jul 15;121(Pt 14):2360-71. doi: 10.1242/jcs.014977. Epub 2008 Jun 24. J Cell Sci. 2008. PMID: 18577581 Free PMC article.
-
Fibronectin polymerization regulates the composition and stability of extracellular matrix fibrils and cell-matrix adhesions.Mol Biol Cell. 2002 Oct;13(10):3546-59. doi: 10.1091/mbc.e02-01-0048. Mol Biol Cell. 2002. PMID: 12388756 Free PMC article.
-
Coordinated regulation of vascular Ca2+ and K+ channels by integrin signaling.Adv Exp Med Biol. 2010;674:69-79. doi: 10.1007/978-1-4419-6066-5_7. Adv Exp Med Biol. 2010. PMID: 20549941 Review.
-
The membrane tethered matrix metalloproteinase MT1-MMP at the forefront of melanoma cell invasion and metastasis.Pharmacol Res. 2016 Sep;111:17-22. doi: 10.1016/j.phrs.2016.05.019. Epub 2016 May 21. Pharmacol Res. 2016. PMID: 27221755 Review.
Cited by
-
Mechanisms and context underlying the role of autophagy in cancer metastasis.Autophagy. 2018;14(7):1110-1128. doi: 10.1080/15548627.2018.1450020. Epub 2018 Jun 4. Autophagy. 2018. PMID: 29863947 Free PMC article. Review.
-
MMP14 is required for delamination of chick neural crest cells independently of its catalytic activity.Development. 2020 Apr 12;147(7):dev183954. doi: 10.1242/dev.183954. Development. 2020. PMID: 32280063 Free PMC article.
-
Matrix metalloproteinase 14 modulates signal transduction and angiogenesis in the cornea.Surv Ophthalmol. 2016 Jul-Aug;61(4):478-97. doi: 10.1016/j.survophthal.2015.11.006. Epub 2015 Dec 2. Surv Ophthalmol. 2016. PMID: 26647161 Free PMC article. Review.
-
Redox-active endosomes mediate α5β1 integrin signaling and promote chondrocyte matrix metalloproteinase production in osteoarthritis.Sci Signal. 2023 Oct 31;16(809):eadf8299. doi: 10.1126/scisignal.adf8299. Epub 2023 Oct 31. Sci Signal. 2023. PMID: 37906629 Free PMC article.
-
Differential Fibrotic Response of Muscle Fibroblasts, Myoblasts, and Myotubes to Cholic and Deoxycholic Acids.Adv Exp Med Biol. 2023;1408:219-234. doi: 10.1007/978-3-031-26163-3_12. Adv Exp Med Biol. 2023. PMID: 37093430
References
-
- Alfranca A., Lopez-Oliva J. M., Genis L., Lopez-Maderuelo D., Mirones I., Salvado D., Quesada A. J., Arroyo A. G., Redondo J. M. (2008). PGE2 induces angiogenesis via MT1-MMP-mediated activation of the TGFbeta/Alk5 signaling pathway. Blood 112, 1120-1128 - PubMed
-
- Barilla M. L., Carsons S. E. (2000). Fibronectin fragments and their role in inflammatory arthritis. Semin. Arthritis Rheum. 29, 252-265 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

