Methods to decrease blood loss and transfusion requirements for liver transplantation
- PMID: 22161443
- PMCID: PMC8939250
- DOI: 10.1002/14651858.CD009052.pub2
Methods to decrease blood loss and transfusion requirements for liver transplantation
Abstract
Background: Excessive blood loss and increased blood transfusion requirements may have significant impact on the short-term and long-term outcomes after liver transplantation.
Objectives: To compare the potential benefits and harms of different methods of decreasing blood loss and blood transfusion requirements during liver transplantation.
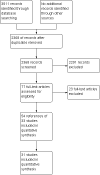
Search methods: We searched The Cochrane Central Register of Controlled Trials in The Cochrane Library, MEDLINE, EMBASE, Science Citation Index Expanded, and metaRegister of Controlled Trials until September 2011.
Selection criteria: We included all randomised clinical trials that were performed to compare various methods of decreasing blood loss and blood transfusion requirements during liver transplantation.
Data collection and analysis: Two authors independently identified the trials and extracted the data. We analysed the data with both the fixed-effect and the random-effects model using RevMan Analysis. For each outcome we calculated the risk ratio (RR), mean difference (MD), or standardised mean difference (SMD) with 95% confidence intervals (CI) based on available data analysis. We also conducted network meta-analysis.
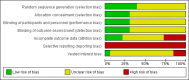
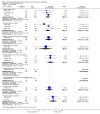
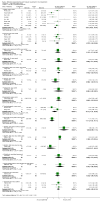
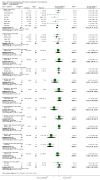
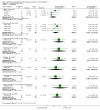
Main results: We included 33 trials involving 1913 patients. The sample size in the trials varied from 8 to 209 participants. The interventions included pharmacological interventions (aprotinin, tranexamic acid, epsilon amino caproic acid, antithrombin 3, recombinant factor (rFvIIa), oestrogen, prostaglandin, epinephrine), blood substitutes (blood components rather than whole blood, hydroxy-ethyl starch, thromboelastography), and cardiovascular interventions (low central venous pressure). All the trials were of high risk of bias. Primary outcomes were reported in at least two trials for the following comparisons: aprotinin versus control, tranexamic acid versus control, recombinant factor VIIa (rFVIIa) versus control, and tranexamic acid versus aprotinin. There were no significant differences in the 60-day mortality (3 trials; 6/161 (3.7%) in the aprotinin group versus 8/119 (6.7%) in the control group; RR 0.52; 95% CI 0.18 to 1.45), primary graft non-function (2 trials; 0/128 (0.0%) in the aprotinin group versus 4/89 (4.5%) in the control group; RR 0.15; 95% CI 0.02 to 1.25), retransplantation (3 trials; 2/256 (0.8%) in the aprotinin group versus 12/178 (6.7%) in the control group; RR 0.21; 95% CI 0.02 to 1.79), or thromboembolic episodes (3 trials; 4/161 (2.5%) in the aprotinin group versus 5/119 (4.2%) in the control group; RR 0.59; 95% CI 0.19 to 1.84) between the aprotinin and control groups. There were no significant differences in the 60-day mortality (3 trials; 4/83 (4.8%) in the tranexamic acid group versus 5/56 (8.9%) in the control group; RR 0.55; 95% CI 0.17 to 1.76), retransplantation (2 trials; 3/41 (7.3%) in the tranexamic acid group versus 3/36 (8.3%) in the control group; RR 0.79; 95% CI 0.18 to 3.48), or thromboembolic episodes (5 trials; 5/103 (4.9%) in the tranexamic acid group versus 1/76 (1.3%) in the control group; RR 2.20; 95% CI 0.38 to 12.64) between the tranexamic acid and control groups. There were no significant differences in the 60-day mortality (3 trials; 8/195 (4.1%) in the recombinant factor VIIa (rFVIIa) group versus 2/91 (2.2%) in the control group; RR 1.51; 95% CI 0.33 to 6.95), thromboembolic episodes (2 trials; 24/185 (13.0%) in the rFVIIa group versus 8/81 (9.9%) in the control group; RR 1.38; 95% CI 0.65 to 2.91), or serious adverse events (2 trials; 90/185 (48.6%) in the rFVIIa group versus 30/81 (37.0%) in the control group; RR 1.30; 95% CI 0.94 to 1.78) between the rFVIIa and control groups. There were no significant differences in the 60-day mortality (2 trials; 6/91 (6.6%) in the tranexamic acid group versus 1/87 (1.1%) in the aprotinin group; RR 4.12; 95% CI 0.71 to 23.76) or thromboembolic episodes (2 trials; 4/91 (4.4%) in the tranexamic acid group versus 2/87 (2.3%) in the aprotinin group; RR 1.97; 95% CI 0.37 to 10.37) between the tranexamic acid and aprotinin groups. The remaining outcomes in the above comparisons and the remaining comparisons included only only trial under the primary outcome or the outcome was not reported at all in the trials. There were no significant differences in the mortality, primary graft non-function, graft failure, retransplantation, thromboembolic episodes, or serious adverse events in any of these comparisons. However, the confidence intervals were wide, and it is not possible to reach any conclusion on the safety of the interventions. None of the trials reported the quality of life in patients.Secondary outcomes were reported in at least two trials for the following comparisons - aprotinin versus control, tranexamic acid versus control, rFVIIa versus control, thromboelastography versus control, and tranexamic acid versus aprotinin. There was significantly lower allogeneic blood transfusion requirements in the aprotinin group than the control group (8 trials; 185 patients in aprotinin group and 190 patients in control group; SMD -0.61; 95% CI -0.82 to -0.40). There were no significant differences in the allogeneic blood transfusion requirements between the tranexamic acid and control groups (4 trials; 93 patients in tranexamic acid group and 66 patients in control group; SMD -0.27; 95% CI -0.59 to 0.06); rFVIIa and control groups (2 trials; 141 patients in rFVIIa group and 80 patients in control group; SMD -0.05; 95% CI -0.32 to 0.23); thromboelastography and control groups (2 trials; 31 patients in thromboelastography group and 31 patients in control group; SMD -0.73; 95% CI -1.69 to 0.24); or between the tranexamic acid and aprotinin groups (3 trials; 101 patients in tranexamic acid group and 97 patients in aprotinin group; SMD -0.09; 95% CI -0.36 to 0.19). The remaining outcomes in the above comparisons and the remaining comparisons included only only trial under the primary outcome or the outcome was not reported at all in the trials. There were no significant differences in the blood loss, transfusion requirements, hospital stay, or intensive care unit stay in most of the comparisons.
Authors' conclusions: Aprotinin, recombinant factor VIIa, and thromboelastography groups may potentially reduce blood loss and transfusion requirements. However, risks of systematic errors (bias) and risks of random errors (play of chance) hamper the confidence in this conclusion. We need further well-designed randomised trials with low risk of systematic error and low risk of random errors before these interventions can be supported or refuted.
Conflict of interest statement
None.
Figures














Update of
References
References to studies included in this review
Ahn 2008 {published data only}
-
- Ahn HJ, Yang M, Gwak MS, Koo MS, Bang SR, Kim GS, et al. Coagulation and biochemical effects of balanced salt‐based high molecular weight vs saline‐based low molecular weight hydroxyethyl starch solutions during the anhepatic period of liver transplantation. Anaesthesia 2008;63(3):235‐42. - PubMed
Baudo 1992 {published data only}
-
- Baudo F, DeGasperi A, deCataldo F, Caimi TM, Cattaneo D, Redaelli R, et al. Antithrombin III supplementation during orthotopic liver transplantation in cirrhotic patients: a randomized trial. Thrombosis Research 1992;68(4‐5):409‐16. - PubMed
Boylan 1996 {published data only}
-
- Boylan JF, Klinck JR, Sandler AN, Arellano R, Greig PD, Nierenberg H, et al. Tranexamic acid reduces blood loss, transfusion requirements, and coagulation factor use in primary orthotopic liver transplantation. Anesthesiology 1996;85(5):1043‐8. - PubMed
Cottam 1991 {published data only}
-
- Cottam S, Hunt B, Segal H, Ginsburg R, Potter D. Aprotinin inhibits tissue plasminogen activator‐mediated fibrinolysis during orthotopic liver transplantation. Transplantation Proceedings 1991;23(3):1933. - PubMed
-
- Hunt B J, Cottam S, Segal H, Ginsburg R, Potter D. Inhibition by aprotinin of tPA‐mediated fibrinolysis during orthotopic liver transplantation. Lancet 1990;336(8711):381. - PubMed
Dalmau 2000 {published data only}
-
- Dalmau A, Sabate A, Acosta F, Garcia Huete L, Koo M, Sansano T, et al. Tranexamic acid reduces red cell transfusion better than epsilon‐ aminocaproic acid or placebo in liver transplantation. Anesthesia and Analgesia 2000;91(1):29‐34. - PubMed
-
- Dalmau A, Sabaté A, Acosta F, Garcia‐Huete L, Koo M, Reche M, et al. Comparative study of antifibrinolytic drugs in orthotopic liver transplantation. Transplantation proceedings 1999;31(6):2361‐2. - PubMed
Dalmau 2004 {published data only}
-
- Dalmau A, Sabaté A, Koo M, Bartolomé C, Rafecas A, Figueras J, et al. The prophylactic use of tranexamic acid and aprotinin in orthotopic liver transplantation: A comparative study. Liver Transplantation 2004;10(2):279‐84. - PubMed
Feng 2010 {published data only}
-
- Feng ZY, Xu X, Zhu SM, Bein B, Zheng SS. Effects of low central venous pressure during preanhepatic phase on blood loss and liver and renal function in liver transplantation. World Journal of Surgery 2010;34(8):1864‐73. - PubMed
Findlay 2001 {published data only}
-
- Findlay JY, Kufner RP. Aprotinin reduces vasoactive medication use during adult liver transplantation. Journal of Clinical Anesthesia 2003;15(1):19‐23. - PubMed
-
- Findlay JY, Rettke SR, Ereth MH, Plevak DJ, Krom RAF, Kufner RP. Aprotinin reduces red blood cell transfusion in orthotopic liver transplantation: A prospective, randomized, double‐blind study. Liver Transplantation 2001;7(9):802‐7. - PubMed
Frenette 1998 {published data only}
-
- Frenette L, Cox J, McArdle P, Eckhoff D, Bynon S. Conjugated estrogen reduces transfusion and coagulation factor requirements in orthotopic liver transplantation. Anesthesia and Analgesia 1998;86(6):1183‐6. - PubMed
Garcia‐Huete 1997 {published data only}
-
- Garcia‐Huete L, Domenech P, Sabate A, MartinezBrotons F, Jaurrieta E, Figueras J. The prophylactic effect of aprotinin on intraoperative bleeding in liver transplantation: A randomized clinical study. Hepatology 1997;26(5):1143‐8. - PubMed
-
- Garcia‐Huete L, Sabate‐Pes A. Aprotinine in orthotopic liver transplantation. Medicina Clinica 1997;108(11):439. - PubMed
-
- García L, Sabaté A, Domenech P, Martinez Brotons F, Drudis R, Jaurrieta E. Aprotinin in orthotopic liver transplantation. Transplantation Proceedings 1995;27(4):2290‐1. - PubMed
Groh 1993 {published data only}
-
- Azad SC, Groh J, Welte M, Pratschke E, Kratzer MAA. Effect of aprotinin on transfusion requirements and coagulation parameters in orthotopic liver transplantation (OLT). Annals of Hematology 1992;64(Supplement):A41.
-
- Groh J, Welte M, Azad S C, Anthuber M, Haller M, Kratzer MA. Does aprotinin really reduce blood loss in orthotopic liver transplantation?. Seminars in Thrombosis and Hemostasis 1993;19(3):306‐8. - PubMed
-
- Groh J, Welte M, Azad SC, Forst H, Pratschke E, Kratzer MAA. Does aprotinin affect blood loss in liver transplantation. Lancet 1992;340(8812):173. - PubMed
-
- Kratzer MA, Azad SC, Groh J, Welte M, Haller M, Pratschke E. [The effects of aprotinin. Blood loss and coagulation parameters in orthotopic liver transplantation: A clinical‐experimental, prospective and randomized double‐blind study]. Der Anaesthesist 1997;46(4):294‐302. - PubMed
-
- Welte M, Groh J, Azad S, Anbuther M, Haller M, Kratzer MAA. No beneficial effect of aprotinin on blood loss and coagulation in liver transplantation. Anesthesiology 1992;77(Supplement 3A):A225.
Hei 2005 {published data only}
-
- Hei ZQ, Luo CF, Li SR, Ma WH, Luo GJ, Chi XJ. Effect of aprotinin on perioperative requirements of blood and liquid in severe hepatitis patients undergoing liver transplantation. Chinese Pharmaceutical Journal 2005;40(18):1432‐5.
Himmelreich 1992 {published data only}
-
- Bechstein WO, Riess H, Blumhardt G, Himmelreich G, Jochum M, Gerlach H, et al. Aprotinin in orthotopic liver transplantation. Seminars in Thrombosis and Hemostasis 1993;19(3):262‐7. - PubMed
-
- Himmelreich G, Bechstein WO, Jochum M, Gerlach H, Neuhaus P, Riess H. Effect of two different dosages of aprotinin on hemostasis and graft function in orthotopic liver transplantation (OLT). Annals of Hematology 1994;68(Suppl 1):A48.
-
- Himmelreich G, Muser M, Neuhaus P, Bechstein WO, Slama KJ, Jochum M, et al. Different aprotinin applications influencing hemostatic changes in orthotopic liver transplantation. Transplantation 1992;53(1):132‐6. - PubMed
Himmelreich 1993 {published data only}
-
- Himmelreich G, Hundt K, Bechstein WO, Rossaint R, Neuhaus P, Riess H. Influence of prostaglandin E1 infusion on hemostasis in orthotopic liver transplantation. Seminars in Thrombosis and Hemostasis 1993;19(3):273‐8. - PubMed
-
- Himmelreich G, Hundt K, Neuhaus P, Bechstein WO, Roissant R, Riess H. Evidence that intraoperative prostaglandin E1 infusion reduces impaired platelet aggregation after reperfusion in orthotopic liver transplantation. Transplantation 1993;55(4):819‐26. - PubMed
Ickx 1993 {published data only}
-
- Ickx B, Pradier O, Groote F, Hendrice C, Gelin M, Stadt TJ, et al. Does aprotinin really reduce blood loss during orthotopic liver transplantation [abstract]. Hepatology 1993;18(3):740.
Ickx 1995 {published data only}
-
- Ickx B, Pierre S, Pradier O, DeGroote F, Vandestadt J, Gelin M, et al. Comparison of the beneficial effect of aprotinin and tranexamic acid on perioperative blood loss during liver transplantation (abstract). British Journal of Anaesthesia 1995;74(Suppl 1):62.
Ickx 2006 {published data only}
-
- Ickx BE, Linden PJ, Melot C, Wijns W, Pauw L, Vandestadt J, et al. Comparison of the effects of aprotinin and tranexamic acid on blood loss and red blood cell transfusion requirements during the late stages of liver transplantation. Transfusion 2006;46(4):595‐605. - PubMed
Kaspar 1997 {published data only}
-
- Kaspar M, Ramsay MAE, Nguyen AT, Cogswell M, Hurst G, Ramsay KJ. Continuous small‐dose tranexamic acid reduces fibrinolysis but not transfusion requirements during orthotopic liver transplantation. Anesthesia and Analgesia 1997;85(2):281‐5. - PubMed
Laine 2003 {published data only}
-
- Laine E, Steadman R, Calhoun L, Blackall D, Levin P, Braunfeld M, et al. Comparison of RBCs and FFP with whole blood during liver transplant surgery. Transfusion 2003;43(3):322‐7. - PubMed
Lassale 1996 {published data only}
-
- Lassale B, Rathelot P, Angelini B, Bongrand MC, Timon David P. Aprotinin in orthotopic liver transplantation (OLT): Clinical and economical interest in blood requirements. Journal de Pharmacie Clinique 1996;15(3):187‐91.
Lodge 2005 {published data only}
-
- Lodge JP, Jonas S, Jones R M, Olausson M, Mir‐Pallardo J, Soefelt S, et al. Efficacy and safety of repeated perioperative doses of recombinant factor VIIa in liver transplantation. Liver Transplantation 2005;11(8):973‐9. - PubMed
-
- Lodge JPA, Jonas S, Jones RM, Olausson M, Mir‐Pallardo J, Soefelt S, et al. Efficcy and safety of recombinant factor VIIa (RFVIIa) on transfusion reduction in orthotopic liver transplantation (OLT) ‐ a randomised, double‐blind, placebo‐controlled trial. Transplantation 2004;78(2):93.
Marcel 1996 {published data only}
-
- Marcel RJ, Stegall WC, Suit CT, Arnold JC, Vera RL, Ramsay MAE, et al. Continuous small‐dose aprotinin controls fibrinolysis during orthotopic liver transplantation. Anesthesia and Analgesia 1996;82(6):1122‐5. - PubMed
Merle 1997 {published data only}
-
- Merle JC, Cherqui D, Lauzet JY, Malassagne B, Duvoux C, Fagniez PL, et al. Aprotinin significantly reduces blood transfusion requirements in liver transplantation for severe cirrhosis: a double‐blind randomized controlled trial. Anesthesiology 1997;87(3A):A83.
Milroy 1995 {published data only}
-
- Milroy SJ, Cottam S, Tan KC, Hilmi I, Oyesola B. Improved haemodynamic stability with administration of aprotinin during orthotopic liver transplantation. British Journal of Anaesthesia 1995;75(6):747‐51. - PubMed
-
- Segal HC, Hunt BJ, Cottam S, Beard C, Francis J L, Potter D, et al. Changes in the contact system during orthotopic liver transplantation with and without aprotinin. Transplantation 1995;59(3):366‐70. - PubMed
-
- Segal HC, Hunt BJ, Cottam S, Downing A, Beard C, Francis JL, et al. Fibrinolytic activity during orthotopic liver transplantation with and without aprotinin. Transplantation 1994;58(12):1356‐60. - PubMed
Planinsic 2005 {published data only}
-
- Planinsic RM, Meer J, Testa G, Grande L, Candela A, Porte RJ, et al. Safety and efficacy of a single bolus administration of recombinant factor VIIa in liver transplantation due to chronic liver disease. Liver Transplantation 2005;11(8):895‐900. - PubMed
Ponnudurai 2005 {published data only}
-
- Ponnudurai RN, Koneru B, Akhtar SA, Wachsberg RH, Fisher A, Wilson DJ, et al. Vasopressor administration during liver transplant surgery and its effect on endotracheal reintubation rate in the postoperative period: a prospective, randomized, double‐blind, placebo‐controlled trial. Clinical Therapeutics 2005;27(2):192‐8. - PubMed
Porte 2000 {published data only}
-
- Molenaar IQ, Begliomini B, Grazi GL, Ringers J, Terpstra OT, Porte RJ. The effect of aprotinin on renal function in orthotopic liver transplantation. Transplantation 2001;71(2):247‐52. - PubMed
-
- Molenaar IQ, Begliomini B, Martinelli G, Putter H, Terpstra OT, Porte RJ. Reduced need for vasopressors in patients receiving aprotinin during orthotopic liver transplantation. Anesthesiology 2001;94(3):433‐8. - PubMed
-
- Molenaar IQ, Legnani C, Groenland TH, Palareti G, Begliomini B, Terpstra OT, et al. Aprotinin in orthotopic liver transplantation: evidence for a prohemostatic, but not a prothrombotic, effect. Liver Transplantation 2001;7(10):896‐903. - PubMed
-
- Porte RJ, Molenaar IQ, Begliomini B, Groenland THN, Januszkiewicz A, Lindgren L, et al. Aprotinin and transfusion requirements in orthotopic liver transplantation: A multicentre randomised double‐blind study. Lancet 2000;355(9212):1303‐9. - PubMed
-
- Porte RJ, Molenaar IQ, Begliomini B, Groenland THN, Januszkiewicz A, Lindgren L, et al. Aprotinin reduces blood loss and transfusion requirements in orthotopic liver transplantation: A placebo‐controlled multicenter study. Transplantation 1999;67(7):S260.
Pugliese 2007 {published data only}
-
- Pugliese F, Ruberto F, Summonti D, Perrella S, Cappannoli A, Tosi A, et al. Activated recombinant factor VII in orthotopic liver transplantation. Transplantation Proceedings 2007;39(6):1883‐5. - PubMed
Rummo 2010 {published data only}
-
- Rummo OO, Shcherba AE, Minou AF, Dzyadzko AM, Avdey EL, Santotski EO, et al. Impact of thromboelastometry on requirement of blood transfusion and incidence of septic complications in deceased donor liver transplantation. Liver Transplantation 2010;16(Suppl 1):S219.
Soilleux 1995 {published data only}
-
- Soilleux H, Gillon MC, Mirand A, Daibes M, Leballe F, Ecoffey C. Comparative effects of small and large aprotinin doses on bleeding during orthotopic liver transplantation. Anesthesia and Analgesia 1995;80(2):349‐52. - PubMed
Wang 2010 {published data only}
-
- Wang SC, Shieh JF, Chang KY, Chu YC, Liu CS, Loong CC, et al. Thromboelastography‐guided transfusion decreases intraoperative blood transfusion during orthotopic liver transplantation: randomized clinical trial. Transplantation Proceedings 2010;42(7):2590‐3. - PubMed
Williamson 1999 {published data only}
-
- Freeman JW, Williamson LM, Llewelyn C, Fisher N, Allain JP, Bellamy M, et al. A randomized trial of solvent/detergent and standard fresh frozen plasma in the treatment of the coagulopathy seen during orthotopic liver transplantation. Vox Sanguinis 1998;74(Supplement 1):225‐9. - PubMed
-
- Williamson LM, Llewelyn CA, Fisher NC, Allain JP, Bellamy MC, Baglin TP, et al. A randomized trial of solvent/detergent‐treated and standard fresh‐frozen plasma in the coagulopathy of liver disease and liver transplantation. Transfusion 1999;39(11‐12):1227‐34. - PubMed
Yassen 1993 {published data only}
-
- Yassen K, Bellamy MC, Sadek SA, Webster NR. Tranexamic acid reduces blood loss during orthotopic liver transplantation. Clinical Transplantation 1993;7(5):453‐8.
References to studies excluded from this review
Chapman 2006 {published data only}
-
- Chapman WC, Lockstadt H, Singla N, Kafie FE, Lawson J H. Phase 2, randomized, double‐blind, placebo‐controlled, multicenter clinical evaluation of recombinant human thrombin in multiple surgical indications. Journal of Thrombosis and Haemostasis 2006;4(9):2083‐5. - PubMed
Grosse 1991 {published data only}
-
- Grosse H, Lobbes W, Frambach M, Broen O, Ringe B, Barthels M. The use of high dose aprotinin in liver transplantation: the influence on fibrinolysis and blood loss. Thrombosis Research 1991;63(3):287‐97. - PubMed
Grosse 1993 {published data only}
-
- Grosse H, Lobbes W, Frambach M, Ringe B, Barthels M. Influence of high‐dose aprotinin on hemostasis and blood requirement in orthotopic liver transplantation. Seminars in Thrombosis and Hemostasis 1993;19(3):302‐5. - PubMed
Ickx 1993a {published data only}
-
- Ickx B, Pradier O, DeGroote F, Hendrice C, Toungouz M, Vandestadt J, et al. Effect of two different dosages of aprotonin on perioperative blood loss during liver transplantation. Seminars in Thrombosis and Hemostasis 1993;19(3):300‐1. - PubMed
Kang 1993 {published data only}
-
- Kang Y. Clinical use of synthetic antifibrinolytic agents during liver transplantation. Seminars in Thrombosis and Hemostasis 1993;19(3):258‐61. - PubMed
Kufner 2000 {published data only}
-
- Kufner R P. Use of antifibrinolytics in orthotopic liver transplantation. Transplantation Proceedings 2000;32(3):636‐7. - PubMed
Lentschener 1996 {published data only}
-
- Lentschener C, Benhamou D, Cottam S, Milroy SJ. Aprotinin during liver transplantation (3). British Journal of Anaesthesia 1996;76(6):882. - PubMed
Levy 2008 {published data only}
-
- Levy JH. Pharmacologic methods to reduce perioperative bleeding. Transfusion 2008;48(1):31S‐38S. - PubMed
Marino 1988 {published data only}
Meijer 2003 {published data only}
-
- Meijer K, Hendriks HG, Wolf JT, Klompmaker IJ, Lisman T, Hagenaars AA, et al. Recombinant factor VIIa in orthotopic liver transplantation: influence on parameters of coagulation and fibrinolysis. Blood Coagulation and Fibrinolysis 2003;14(2):169‐74. - PubMed
Molenaar 2002 {published data only}
-
- Molenaar IQ, Porte RJ. Aprotinin and thromboembolism in liver transplantation: Is there really a causal effect?. Anesthesia and Analgesia 2002;94(5):1367‐8. - PubMed
Neuhaus 1989 {published data only}
-
- Neuhaus P, Bechstein WO, Lefèbre B, Blumhardt G, Slama K. Effect of aprotinin on intraoperative bleeding and fibrinolysis in liver transplantation. Lancet 1989;335(8668):924‐5. - PubMed
Palareti 1991 {published data only}
-
- Palareti G, Legnani C, Maccaferri M, Gozzetti G, Mazziotti A, Martinelli G, et al. Coagulation and fibrinolysis in orthotopic liver transplantation: role of the recipient's disease and use of antithrombin III concentrates. S. Orsola Working Group on Liver Transplantation. Haemostasis 1991;21(2):68‐76. - PubMed
Porte 2001 {published data only}
-
- Porte RJ, Slooff MJH. Aprotinin: Safe and effective in all patients undergoing orthotopic liver transplantation?. Liver Transplantation 2001;7(9):808‐10. - PubMed
Porte 2004 {published data only}
-
- Porte RJ. Antifibrinolytics in liver transplantation: They are effective, but what about the risk‐benefit ratio?. Liver Transplantation 2004;10(2):285‐8. - PubMed
Porte 2004a {published data only}
-
- Porte RJ, Hendriks HG, Slooff MJ. Blood conservation in liver transplantation: The role of aprotinin. Journal of Cardiothoracic and Vascular Anesthesia 2004;18(4 Suppl):31S‐37S. - PubMed
Porte 2005 {published data only}
-
- Porte RJ, Caldwell SH. The role of recombinant factor VIIa in liver transplantation. Liver Transplantation 2005;11(8):872‐4. - PubMed
Rosea 2000 {published data only}
-
- Rosea E. Liver transplantation, the problem of peroperative bleeding. Presse Medicale 2000;29(23):1289‐90. - PubMed
Sankarankutty 2006 {published data only}
-
- Sankarankutty AK, Teixeira AC, Souza FF, Mente ED, Oliveira GR, Almeida RC, et al. Impact of blood salvage during liver transplantation on reduction in transfusion requirements. Acta Cirurgica Brasileira 2006;21(Suppl 1):44‐7. - PubMed
Segal 2000 {published data only}
-
- Segal H, Hunt BJ. Aprotinin: pharmacological reduction of perioperative bleeding. Lancet 2000;355(9212):1289‐90. - PubMed
Spohr 2002 {published data only}
Suárez 1993 {published data only}
-
- Suárez M, Sangro B, Herrero JI, Picardi A, Páramo JA, Quiroga J, et al. Effectiveness of aprotinin in orthotopic liver transplantation. Seminars in Thrombosis and Hemostasis 1993;19(3):292‐6. - PubMed
Additional references
Bismuth 1987
-
- Bismuth H, Castaing D, Ericzon BG, Otte JB, Rolles K, Ringe B, et al. Hepatic transplantation in Europe. First Report of the European Liver Transplant Registry. Lancet 1987;2(8560):674‐6. - PubMed
Boin 2008
-
- Boin IF, Leonardi MI, Luzo AC, Cardoso AR, Caruy CA, Leonardi LS. Intraoperative massive transfusion decreases survival after liver transplantation. Transplantation Proceedings 2008;40(3):789‐91. - PubMed
Boyd 2007
-
- Boyd SD, Stenard F, Lee DK, Goodnough LT, Esquivel CO, Fontaine MJ. Alloimmunization to red blood cell antigens affects clinical outcomes in liver transplant patients. Liver Transplantation 2007;13(12):1654‐61. - PubMed
Calne 1979
-
- Calne RY, Smith DP, McMaster P, Craddock GN, Rolles K, Farman JV, et al. Use of partial cardiopulmonary bypass during the anhepatic phase of orthotopic liver grafting. Lancet 1979;2(8143):612‐4. - PubMed
Cescon 2006
-
- Cescon M, Grazi GL, Grassi A, Ravaioli M, Vetrone G, Ercolani G, et al. Effect of ischemic preconditioning in whole liver transplantation from deceased donors. A pilot study. Liver Transplantation 2006;12(4):628‐35. - PubMed
Corno 2006
-
- Corno V, Colledan M, Dezza MC, Guizzetti M, Lucianetti A, Maldini G, et al. Extended right split liver graft for primary transplantation in children and adults. Transplantation International 2006;19(6):492‐9. - PubMed
DeMets 1987
-
- DeMets DL. Methods for combining randomized clinical trials: strengths and limitations. Statistics in Medicine 1987;6(3):341‐50. - PubMed
DerSimonian 1986
-
- DerSimonian R, Laird N. Meta‐analysis in clinical trials. Controlled Clinical Trials 1986;7(3):177‐88. - PubMed
Diprose 2005
-
- Diprose P, Herbertson MJ, O'Shaughnessy D, Deakin CD, Gill RS. Reducing allogeneic transfusion in cardiac surgery: A randomized double‐blind placebo‐controlled trial of antifibrinolytic therapies used in addition to intra‐operative cell salvage. British Journal of Anaesthesia 2005;94(3):271‐8. - PubMed
Egger 1997
ELTR 2011
-
- ELTR. European liver transplant registry. Evolution of 93,364 liver transplantations in Europe. http://www.eltr.org/spip.php?article152 2011 (accessed on 29th August 2011).
Fergusson 2008
-
- Fergusson DA, Hebert PC, Mazer CD, Fremes S, MacAdams C, Murkin JM, et al. A comparison of aprotinin and lysine analogues in high‐risk cardiac surgery. New England Journal of Medicine 2008;358(22):2319‐31. - PubMed
Gluud 2011
-
- Gluud C, Nikolova D, Klingenberg SL, Alexakis N, Als‐Nielsen B, Colli A, et al. Cochrane Hepato‐Biliary Group. About The Cochrane Collaboration (Cochrane Review Groups (CRGs)). 2011, Issue 2. Art. No.: LIVER.
Gurusamy 2009a
Gurusamy 2009b
Gurusamy 2009c
-
- Gurusamy KS, Gluud C, Nikolova D, Davidson BR. Assessment of risk of bias in randomized clinical trials in surgery. The British Journal of Surgery 2009;96(4):342‐9. - PubMed
Gurusamy 2011a
Gurusamy 2011b
Hendriks 2005
-
- Hendriks HG, Meer J, Wolf JT, Peeters PM, Porte RJ, Jong K, et al. Intraoperative blood transfusion requirement is the main determinant of early surgical re‐intervention after orthotopic liver transplantation. Transplant International 2005;17(11):673‐9. - PubMed
Henry 2011
Higgins 2002
-
- Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta‐analysis. Statistics in Medicine 2002;21(11):1539‐58. - PubMed
Higgins 2011
-
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Colloboration, 2011. Available from www.cochrane‐handbook.org.
ICH‐GCP 1997
-
- International Conference on Harmonisation Expert Working Group. International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. ICH harmonised tripartite guideline. Guideline for good clinical practice1997 CFR & ICH Guidelines. Vol. 1, PA 19063‐2043, USA: Barnett International/PAREXEL, 1997.
Jabbour 2005
-
- Jabbour N, Gagandeep S, Mateo R, Sher L, Genyk Y, Selby R. Transfusion free surgery: single institution experience of 27 consecutive liver transplants in Jehovah's Witnesses. Journal of the American College of Surgeons 2005;201(3):412‐7. - PubMed
Jeon 2010
-
- Jeon H, Lee SG. Living donor liver transplantation. Current Opinion in Organ Transplantation 2010;15(3):283‐7. - PubMed
Kamath 2001
-
- Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, et al. A model to predict survival in patients with end‐stage liver disease. Hepatology 2001;33(2):464‐70. - PubMed
Kjaergard 2001
-
- Kjaergard LL, Villumsen J, Gluud C. Reported methodologic quality and discrepancies between large and small randomized trials in meta‐analyses. Annals of Internal Medicine 2001;135(11):982‐9. - PubMed
Koneru 2005
-
- Koneru B, Fisher A, He Y, Klein KM, Skurnick J, Wilson DJ, et al. Ischemic preconditioning in deceased donor liver transplantation: a prospective randomized clinical trial of safety and efficacy. Liver Transplantation 2005;11(2):196‐202. - PubMed
Lim 2006
-
- Lim SG, Wai CT, Costa M, Sutedja DS, Lee YM, Lee KH, et al. Referral patterns and waiting times for liver transplantation in Singapore. Singapore Medical Journal 2006;47(7):599‐603. - PubMed
Liu 2008
Macaskill 2001
-
- Macaskill P, Walter SD, Irwig L. A comparison of methods to detect publication bias in meta‐analysis. Statistics in Medicine 2001;20(4):641‐54. - PubMed
Massicotte 2005
-
- Massicotte L, Sassine MP, Lenis S, Seal RF, Roy A. Survival rate changes with transfusion of blood products during liver transplantation. Canadian Journal of Anaesthesia 2005;52(2):148‐55. - PubMed
Massicotte 2006
-
- Massicotte L, Lenis S, Thibeault L, Sassine MP, Seal RF, Roy A. Effect of low central venous pressure and phlebotomy on blood product transfusion requirements during liver transplantations. Liver Transplantation 2006;12(1):117‐23. - PubMed
MHRA 2007
-
- MHRA. Aprotinin (Trasylol): Suspension of UK marketing authorisations (licences). http://www.mhra.gov.uk/Safetyinformation/Safetywarningsalertsandrecalls/... 2007 (accessed 3rd September 2011).
Mills 2011
Moher 1998
-
- Moher D, Pham B, Jones A, Cook DJ, Jadad AR, Moher M, et al. Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta‐analyses?. Lancet 1998;352(9128):609‐13. - PubMed
Molenaar 2007
-
- Molenaar IQ, Warnaar N, Groen H, Tenvergert EM, Slooff MJ, Porte RJ. Efficacy and safety of antifibrinolytic drugs in liver transplantation: a systematic review and meta‐analysis. American Journal of Transplantation 2007;7(1):185‐94. - PubMed
Murphy 2004
-
- Murphy GJ, Allen SM, Unsworth‐White J, Lewis CT, Dalrymple‐Hay MJ. Safety and efficacy of perioperative cell salvage and autotransfusion after coronary artery bypass grafting: A randomized trial. Annals of Thoracic Surgery 2004;77(5):1553‐9. - PubMed
Nardo 2005
-
- Nardo B, Bertelli R, Montalti R, Beltempo P, Puviani L, Pacile V, et al. Red blood cell transfusion in liver transplantation: a case‐control study. Transplantation Proceedings 2005;37(10):4389‐92. - PubMed
Newell 1992
-
- Newell DJ. Intention‐to‐treat analysis: implications for quantitative and qualitative research. International Journal of Epidemiology 1992;21(5):837‐41. - PubMed
OPTN/SRTR 2009
-
- OPTN/SRTR. Welcome to the 2009 OPTN / SRTR annual report: Transplant data 1999‐2008. http://www.ustransplant.org/annual_reports/current/default.htm 2009 (accessed on 7th March 2011).
Parmar 1998
-
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta‐analyses of the published literature for survival endpoints. Statistics in Medicine 1998;17(24):2815‐34. - PubMed
Pivalizza 2003
-
- Pivalizza EG, Warters RD, Gebhard R. Desmopressin before liver transplantation. Canadian Journal of Anaesthesia 2003;50(7):748‐9. - PubMed
Ramos 2003
-
- Ramos E, Dalmau A, Sabate A, Lama C, Llado L, Figueras J, et al. Intraoperative red blood cell transfusion in liver transplantation: influence on patient outcome, prediction of requirements, and measures to reduce them. Liver Transplantation 2003;9(12):1320‐7. - PubMed
Ray 2008
-
- Ray WA, Stein CM. The aprotinin story ‐ is BART the final chapter?. New England Journal of Medicine 2008;358(22):2398‐400. - PubMed
RevMan 2011 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.1. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2011.
Royle 2003
-
- Royle P, Milne R. Literature searching for randomized controlled trials used in Cochrane reviews: rapid versus exhaustive searches. International Journal of Technology Assessment in Health Care 2003;19(4):591‐603. - PubMed
Salanti 2008
-
- Salanti G, Higgins JP, Ades AE, Ioannidis JP. Evaluation of networks of randomized trials. Statistical Methods in Medical Research 2008;17(3):279‐301. - PubMed
SAS 9.2 [Computer program]
-
- SAS Institute Inc. SAS 9.2. Cary, NC, USA.: SAS Institute Inc, 2008.
Schulz 1995
-
- Schulz KF, Chalmers I, Hayes RJ, Altman DG. Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. JAMA 1995;273(5):408‐12. - PubMed
Shiffman 2006
-
- Shiffman ML, Saab S, Feng S, Abecassis MI, Tzakis AG, Goodrich NP, et al. Liver and intestine transplantation in the United States, 1995‐2004. American Journal of Transplantation 2006;6(5 Pt 2):1170‐87. - PubMed
StatsDirect 2.7 [Computer program]
-
- StatsDirect Ltd. StatsDirect statistical software. Version 2.7.8. StatsDirect Ltd, 2010.
Thorlund 2011
-
- Thorlund K, Engstrøm J, Wetterslev J, Brok J, Imberger G, Gluud C. Trial sequential analysis (TSA). http://ctu.dk/tsa/downloads.aspx 2011.
Tierney 2007
TSA 2011 [Computer program]
-
- Copenhagen Trial Unit. TSA viewer. Version 0.9 Beta. Copenhagen Trial Unit, 2011.
Wetterslev 2008
-
- Wetterslev J, Thorlund K, Brok J, Gluud C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta‐analysis. Journal of Clinical Epidemiology 2008;61(1):64‐75. - PubMed
Wetterslev 2009
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

