Label-free electrochemical detection of the specific oligonucleotide sequence of dengue virus type 1 on pencil graphite electrodes
- PMID: 22163916
- PMCID: PMC3231433
- DOI: 10.3390/s110605616
Label-free electrochemical detection of the specific oligonucleotide sequence of dengue virus type 1 on pencil graphite electrodes
Abstract
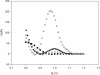
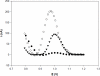
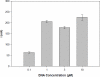
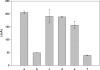
A biosensor that relies on the adsorption immobilization of the 18-mer single-stranded nucleic acid related to dengue virus gene 1 on activated pencil graphite was developed. Hybridization between the probe and its complementary oligonucleotides (the target) was investigated by monitoring guanine oxidation by differential pulse voltammetry (DPV). The pencil graphite electrode was made of ordinary pencil lead (type 4B). The polished surface of the working electrode was activated by applying a potential of 1.8 V for 5 min. Afterward, the dengue oligonucleotides probe was immobilized on the activated electrode by applying 0.5 V to the electrode in 0.5 M acetate buffer (pH 5.0) for 5 min. The hybridization process was carried out by incubating at the annealing temperature of the oligonucleotides. A time of five minutes and concentration of 1 μM were found to be the optimal conditions for probe immobilization. The electrochemical detection of annealing between the DNA probe (TS-1P) immobilized on the modified electrode, and the target (TS-1T) was achieved. The target could be quantified in a range from 1 to 40 nM with good linearity and a detection limit of 0.92 nM. The specificity of the electrochemical biosensor was tested using non-complementary sequences of dengue virus 2 and 3.
Keywords: dengue virus; guanine oxidation; nucleic acid biosensor.
Figures


Similar articles
-
Electrochemical label-free and sensitive nanobiosensing of DNA hybridization by graphene oxide modified pencil graphite electrode.Anal Biochem. 2017 Sep 1;532:64-71. doi: 10.1016/j.ab.2017.06.004. Epub 2017 Jun 9. Anal Biochem. 2017. PMID: 28606426
-
Preparation and characterization of zinc oxide nanoparticles and their sensor applications for electrochemical monitoring of nucleic acid hybridization.Colloids Surf B Biointerfaces. 2011 Sep 1;86(2):397-403. doi: 10.1016/j.colsurfb.2011.04.030. Epub 2011 Apr 30. Colloids Surf B Biointerfaces. 2011. PMID: 21600741
-
Sequence-specific label-free nucleic acid biosensor for the detection of the hepatitis C virus genotype 1a using a disposable pencil graphite electrode.Artif Cells Nanomed Biotechnol. 2016 May;44(3):912-7. doi: 10.3109/21691401.2014.998831. Epub 2015 Jan 24. Artif Cells Nanomed Biotechnol. 2016. PMID: 25619757
-
Surface immobilization strategies for the development of electrochemical nucleic acid sensors.Biosens Bioelectron. 2023 Oct 1;237:115440. doi: 10.1016/j.bios.2023.115440. Epub 2023 Jun 16. Biosens Bioelectron. 2023. PMID: 37406480 Review.
-
Use of Graphene and Its Derivatives for the Detection of Dengue Virus.Biosensors (Basel). 2023 Mar 6;13(3):349. doi: 10.3390/bios13030349. Biosensors (Basel). 2023. PMID: 36979561 Free PMC article. Review.
Cited by
-
Field-Based cDNA-Biosensor for Accurate Detection of Canine Distemper Virus in Tissue Samples.Appl Biochem Biotechnol. 2025 Feb;197(2):1242-1257. doi: 10.1007/s12010-024-05088-x. Epub 2024 Nov 20. Appl Biochem Biotechnol. 2025. PMID: 39565538
-
Diagnostic tests for hepatitis C: recent trends in electrochemical immunosensor and genosensor analysis.World J Gastroenterol. 2014 Nov 14;20(42):15476-91. doi: 10.3748/wjg.v20.i42.15476. World J Gastroenterol. 2014. PMID: 25400433 Free PMC article. Review.
-
Impedimetric DNA biosensor based on a nanoporous alumina membrane for the detection of the specific oligonucleotide sequence of dengue virus.Sensors (Basel). 2013 Jun 17;13(6):7774-85. doi: 10.3390/s130607774. Sensors (Basel). 2013. PMID: 23774989 Free PMC article.
-
A Review of Detection Methods for Vancomycin-Resistant Enterococci (VRE) Genes: From Conventional Approaches to Potentially Electrochemical DNA Biosensors.Biosensors (Basel). 2023 Feb 18;13(2):294. doi: 10.3390/bios13020294. Biosensors (Basel). 2023. PMID: 36832060 Free PMC article. Review.
-
Diagnosis of Dengue Infection Using Conventional and Biosensor Based Techniques.Viruses. 2015 Oct 19;7(10):5410-27. doi: 10.3390/v7102877. Viruses. 2015. PMID: 26492265 Free PMC article. Review.
References
-
- Barrett ADT, Stanberry LR. Dengue. In: Vaughn DW, Whitehead SS, Durbin AP, editors. Vaccines for Biodefense and Emerging and Neglected Diseases. 1st ed. Elsevier; New York, NY, USA: 2009. pp. 288–313.
-
- Araujo JM, Nogueira RM, Schatzmayr HG, Zanotto PM, Bello G. Phylogeography and evolutionary history of dengue virus type 3. Infect. Genet. Evol. 2009;9:716–725. - PubMed
-
- Santos H, Tai MHH, Rocha LFN, Silva HHG, Luz C. Dependence of Metarhizium anisopliae on high humidity for ovicidal activity on Aedes aegypti Adelair. Biol. Contr. 2009;50:37–42.
-
- Ribeiro KA, de Carvalho CM, Molina MT, Lima EP, Lopez-Montero E, Reys JR, de Oliveira MB, Pinto AV, Santana AE, Goulart MO. Activities of naphthoquinones against Aedes aegypti (Linnaeus, 1762) (Diptera: Culicidae), vector of dengue and Biomphalaria glabrata (Say, 1818), intermediate host of Schistosoma mansoni. Acta Tropica. 2009;111:44–50. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

