A novel U-STAT3-dependent mechanism mediates the deleterious effects of chronic nicotine exposure on renal injury
- PMID: 22169004
- PMCID: PMC3311312
- DOI: 10.1152/ajprenal.00338.2011
A novel U-STAT3-dependent mechanism mediates the deleterious effects of chronic nicotine exposure on renal injury
Abstract
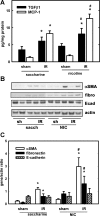
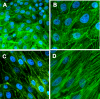
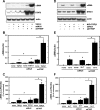
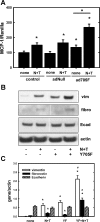
Previous data from our group have demonstrated (Arany I, Grifoni S, Clark JS, Csongradi, Maric C, Juncos LA. Am J Physiol Renal Physiol 301: F125-F133, 2011) that chronic nicotine (NIC) exposure exacerbates acute renal ischemic injury (AKI) in mice that could increase the risk for development and progression of chronic kidney disease (CKD). It has been shown that proximal tubules of the kidney can acquire characteristics that may compromise structural recovery and favor development of inflammation and fibrosis following injury. Chronic NIC exposure can amplify this epithelial process although the mechanism is not identified. Recently, the unphosphorylated form of signal transducer and activator of transcription-3 (U-STAT3) has emerged as a noncanonical mediator of inflammation and fibrosis that may be responsible for the effects of chronic NIC. We found that levels of transforming growth factor β-1 (TGF-β1), α-smooth muscle actin (α-SMA), fibronectin, monocyte chemotactic protein-1 (MCP-1), and expression of U-STAT3 were increased in the ischemic kidneys of NIC-exposed mice. Chronic NIC exposure also increased TGF-β1-dependent F-actin reorganization, vimentin, fibronectin, and α-SMA expression as well as promoter activity of α-SMA and MCP-1 without significant loss of epithelial characteristics (E-cadherin) in cultured renal proximal tubule cells. Importantly, transduction of cells with a U-STAT3 mimetic (Y705F-STAT3) augmented stress fiber formation and also amplified NIC+TGF-β1-induced expression of α-SMA, vimentin, fibronectin, as well as promoter activity of α-SMA and MCP-1. Our results reveal a novel, chronic NIC-exposure-related and U-STAT3-dependent mechanism as mediator of a sustained transcription of genes that are linked to remodeling and inflammation in the kidney during injury. This process may facilitate progression of AKI to CKD. The obtained data may lead to devising therapeutic methods to specifically enhance the protective and/or inhibit adverse effects of STAT3 in the kidney.
Figures


References
-
- Arany I, Megyesi J, Nelkin BD, Safirstein RL. STAT3 attenuates EGFR-mediated ERK activation and cell survival during oxidant stress in mouse proximal tubular cells. Kidney Int 70: 669–674, 2006 - PubMed
-
- Arredondo J, Chernyavsky AI, Jolkovsky DL, Pinkerton KE, Grando SA. Receptor-mediated tobacco toxicity: cooperation of the Ras/Raf-1/MEK1/ERK and JAK-2/STAT-3 pathways downstream of alpha7 nicotinic receptor in oral keratinocytes. FASEB J 20: 2093–2101, 2006 - PubMed
-
- Bottinger EP. TGF-beta in renal injury and disease. Semin Nephrol 27: 309–320, 2007 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

