Vesicular zinc regulates the Ca2+ sensitivity of a subpopulation of presynaptic vesicles at hippocampal mossy fiber terminals
- PMID: 22171030
- PMCID: PMC6623886
- DOI: 10.1523/JNEUROSCI.4164-11.2011
Vesicular zinc regulates the Ca2+ sensitivity of a subpopulation of presynaptic vesicles at hippocampal mossy fiber terminals
Abstract
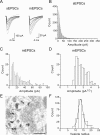
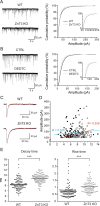
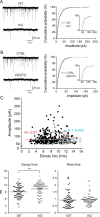
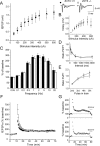
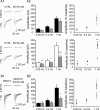
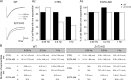
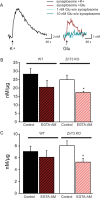
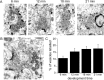
Synaptic vesicles segregate into functionally diverse subpopulations within presynaptic terminals, yet there is no information about how this may occur. Here we demonstrate that a distinct subgroup of vesicles within individual glutamatergic, mossy fiber terminals contain vesicular zinc that is critical for the rapid release of a subgroup of synaptic vesicles during increased activity in mice. In particular, vesicular zinc dictates the Ca(2+) sensitivity of release during high-frequency firing. Intense synaptic activity alters the subcellular distribution of zinc in presynaptic terminals and decreases the number of zinc-containing vesicles. Zinc staining also appears in endosomes, an observation that is consistent with the preferential replenishment of zinc-enriched vesicles by bulk endocytosis. We propose that functionally diverse vesicle pools with unique membrane protein composition support different modes of transmission and are generated via distinct recycling pathways.
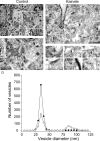
Figures
Similar articles
-
Properties of Hippocampal Mossy Fibre Synapses in VAMP7 KO Mice.Eur J Neurosci. 2025 Feb;61(4):e70016. doi: 10.1111/ejn.70016. Eur J Neurosci. 2025. PMID: 39988820 Free PMC article.
-
Chronic early life lead (Pb2+) exposure alters presynaptic vesicle pools in hippocampal synapses.BMC Pharmacol Toxicol. 2016 Nov 2;17(1):56. doi: 10.1186/s40360-016-0098-1. BMC Pharmacol Toxicol. 2016. PMID: 27802838 Free PMC article.
-
Presynaptic cAMP-PKA-mediated potentiation induces reconfiguration of synaptic vesicle pools and channel-vesicle coupling at hippocampal mossy fiber boutons.PLoS Biol. 2024 Nov 18;22(11):e3002879. doi: 10.1371/journal.pbio.3002879. eCollection 2024 Nov. PLoS Biol. 2024. PMID: 39556620 Free PMC article.
-
RIM1: an edge for presynaptic plasticity.Trends Neurosci. 2002 Jul;25(7):329-32. doi: 10.1016/s0166-2236(02)02193-8. Trends Neurosci. 2002. PMID: 12079752 Review.
-
Timing and efficacy of transmitter release at mossy fiber synapses in the hippocampal network.Pflugers Arch. 2006 Dec;453(3):361-72. doi: 10.1007/s00424-006-0093-2. Epub 2006 Jun 27. Pflugers Arch. 2006. PMID: 16802161 Review.
Cited by
-
Synaptic Zn2+ inhibits neurotransmitter release by promoting endocannabinoid synthesis.J Neurosci. 2013 May 29;33(22):9259-72. doi: 10.1523/JNEUROSCI.0237-13.2013. J Neurosci. 2013. PMID: 23719795 Free PMC article.
-
The Impact of Synaptic Zn2+ Dynamics on Cognition and Its Decline.Int J Mol Sci. 2017 Nov 14;18(11):2411. doi: 10.3390/ijms18112411. Int J Mol Sci. 2017. PMID: 29135924 Free PMC article. Review.
-
AMPA receptor inhibition by synaptically released zinc.Proc Natl Acad Sci U S A. 2015 Dec 22;112(51):15749-54. doi: 10.1073/pnas.1512296112. Epub 2015 Dec 8. Proc Natl Acad Sci U S A. 2015. PMID: 26647187 Free PMC article.
-
Linear-nonlinear cascades capture synaptic dynamics.PLoS Comput Biol. 2021 Mar 15;17(3):e1008013. doi: 10.1371/journal.pcbi.1008013. eCollection 2021 Mar. PLoS Comput Biol. 2021. PMID: 33720935 Free PMC article.
-
Sorting of the vesicular GABA transporter to functional vesicle pools by an atypical dileucine-like motif.J Neurosci. 2013 Jun 26;33(26):10634-46. doi: 10.1523/JNEUROSCI.0329-13.2013. J Neurosci. 2013. PMID: 23804087 Free PMC article.
References
-
- Amaral DG, Witter MP. The three-dimensional organization of the hippocampal formation: a review of anatomical data. Neuroscience. 1989;31:571–591. - PubMed
-
- Aravanis AM, Pyle JL, Harata NC, Tsien RW. Imaging single synaptic vesicles undergoing repeated fusion events: kissing, running, and kissing again. Neuropharmacology. 2003;45:797–813. - PubMed
-
- Augustine GJ. How does calcium trigger neurotransmitter release? Curr Opin Neurobiol. 2001;11:320–326. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
