Intraperitoneal clearance as a potential biomarker of cisplatin after intraperitoneal perioperative chemotherapy: a population pharmacokinetic study
- PMID: 22173671
- PMCID: PMC3273361
- DOI: 10.1038/bjc.2011.557
Intraperitoneal clearance as a potential biomarker of cisplatin after intraperitoneal perioperative chemotherapy: a population pharmacokinetic study
Abstract
Background: Intraperitoneal (IP) perioperative chemotherapy with cisplatin is an interesting option in ovarian cancer treatment. A combination of cisplatin with IP epinephrine (already shown to improve IP and decrease systemic platinum (Pt) exposure) was evaluated using a population pharmacokinetic analysis.
Methods: Data from 55 patients treated with cisplatin-based IP perioperative chemotherapy with (n=26) or without (n=29) epinephrine were analysed using NONMEM.
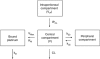
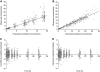
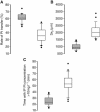
Results: Epinephrine halves clearance between peritoneum and serum (IPCL) and increases the Pt central volume of distribution, IP exposure and penetration in tissue. IPCL has a better predictive value than any other parameter with respect to renal toxicity.
Conclusion: This confirms that IPCL could be useful in assessing renal toxicity. As IPCL is also linked to tissue penetration and IP exposure, it may be proposed as biomarker. In addition to a Bayesian estimation, we propose a single-sample calculation-way to assess it. Prospective studies are needed to validate IPCL as a biomarker in this context.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Aebi S, Castiglione M (2008) Epithelial ovarian carcinoma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 19(Suppl 2): ii14–ii16 - PubMed
-
- Alberts DS, Liu PY, Hannigan EV, O’Toole R, Williams SD, Young JA, Franklin EW, Clarke-Pearson DL, Malviya VK, DuBeshter B (1996) Intraperitoneal cisplatin plus intravenous cyclophosphamide versus intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. N Engl J Med 335: 1950–1955 - PubMed
-
- Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, Lele S, Copeland LJ, Walker JL, Burger RA (2006) Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med 354: 34–43 - PubMed
-
- Beal SL, Sheiner LB, Boeckmann AJ NONMEM Users Guides. Ellicott City MD 1989-2006
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

