Adverse events following 12 and 18 month vaccinations: a population-based, self-controlled case series analysis
- PMID: 22174753
- PMCID: PMC3236196
- DOI: 10.1371/journal.pone.0027897
Adverse events following 12 and 18 month vaccinations: a population-based, self-controlled case series analysis
Abstract
Background: Live vaccines have distinct safety profiles, potentially causing systemic reactions one to 2 weeks after administration. In the province of Ontario, Canada, live MMR vaccine is currently recommended at age 12 months and 18 months.
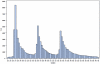
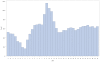
Methods: Using the self-controlled case series design we examined 271,495 12 month vaccinations and 184,312 18 month vaccinations to examine the relative incidence of the composite endpoint of emergency room visits or hospital admissions in consecutive one day intervals following vaccination. These were compared to a control period 20 to 28 days later. In a post-hoc analysis we examined the reasons for emergency room visits and the average acuity score at presentation for children during the at-risk period following the 12 month vaccine.
Results: Four to 12 days post 12 month vaccination, children had a 1.33 (1.29-1.38) increased relative incidence of the combined endpoint compared to the control period, or at least one event during the risk interval for every 168 children vaccinated. Ten to 12 days post 18 month vaccination, the relative incidence was 1.25 (95%, 1.17-1.33) which represented at least one excess event for every 730 children vaccinated. The primary reason for increased events was statistically significant elevations in emergency room visits following all vaccinations. There were non-significant increases in hospital admissions. There were an additional 20 febrile seizures for every 100,000 vaccinated at 12 months.
Conclusions: There are significantly elevated risks of primarily emergency room visits approximately one to two weeks following 12 and 18 month vaccination. Future studies should examine whether these events could be predicted or prevented.
Conflict of interest statement
Figures



References
-
- Stratton KR, Howe CJ, Johnston RB, editors. Adverse Events Associated with Childhood Vaccines: Evidence Bearing on Causality. Washington, DC: National Academy Press; 1994. - PubMed
-
- Siegrist CA. Mechanisms underlying adverse reactions to vaccines. J Comp Pathol. 2007;137(Suppl 1):S46–50. - PubMed
-
- Strebel PM, Papania MJ, Halsey NA. Measles vaccine. In: Plotkin SA, Orenstein WA, editors. Vaccines. 4th ed. Philadelphia: Saunders; 2004.
-
- Fritzell B. Detection of adverse events: what are the current sensitivity limits during clinical development? Vaccine. 2001;20(Suppl 1):S47–48. - PubMed
-
- Barlow WE, Davis RL, Glasser JW, Rhodes PH, Thompson RS, et al. The risk of seizures after receipt of whole-cell pertussis or measles, mumps, and rubella vaccine. N Engl J Med. 2001;345:656–661. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

