XPA-mediated regulation of global nucleotide excision repair by ATR Is p53-dependent and occurs primarily in S-phase
- PMID: 22174788
- PMCID: PMC3236200
- DOI: 10.1371/journal.pone.0028326
XPA-mediated regulation of global nucleotide excision repair by ATR Is p53-dependent and occurs primarily in S-phase
Abstract
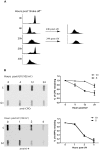
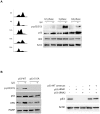
Cell cycle checkpoints play an important role in regulation of DNA repair pathways. However, how the regulation occurs throughout the cell cycle remains largely unknown. Here we demonstrate that nucleotide excision repair (NER) is regulated by the ATR/p53 checkpoint via modulation of XPA nuclear import and that this regulation occurs in a cell cycle-dependent manner. We show that depletion of p53 abrogated the UV-induced nuclear translocation of XPA, while silencing of Chk1 or MAPKAP Kinase-2 (MK2) had no effect. Inhibition of p53 transcriptional activities and silencing of p53-Ser15 phosphorylation also reduced the damage-induced XPA nuclear import. Furthermore, in G1-phase cells the majority of XPA remained in the cytoplasm even after UV treatment. By contrast, while most of the XPA in S-phase cells was initially located in the cytoplasm before DNA damage, UV irradiation stimulated bulk import of XPA into the nucleus. Interestingly, the majority of XPA molecules always were located in the nucleus in G2-phase cells no matter whether the DNA was damaged or not. Consistently, the UV-induced Ser15 phosphorylation of p53 occurred mainly in S-phase cells, and removal of cyclobutane pyrimidine dimers (CPDs) was much more efficient in S-phase cells than in G1-phase cells. Our results suggest that upon DNA damage in S phase, NER could be regulated by the ATR/p53-dependent checkpoint via modulation of the XPA nuclear import process. In contrast, the nuclear import of XPA in G(1) or G(2) phase appears to be largely independent of DNA damage and p53.
Conflict of interest statement
Figures



Similar articles
-
Checkpoint kinase ATR promotes nucleotide excision repair of UV-induced DNA damage via physical interaction with xeroderma pigmentosum group A.J Biol Chem. 2009 Sep 4;284(36):24213-22. doi: 10.1074/jbc.M109.000745. Epub 2009 Jul 8. J Biol Chem. 2009. PMID: 19586908 Free PMC article.
-
ATR-dependent checkpoint modulates XPA nuclear import in response to UV irradiation.Oncogene. 2007 Feb 1;26(5):757-64. doi: 10.1038/sj.onc.1209828. Epub 2006 Jul 24. Oncogene. 2007. PMID: 16862173 Free PMC article.
-
Phosphorylation of nucleotide excision repair factor xeroderma pigmentosum group A by ataxia telangiectasia mutated and Rad3-related-dependent checkpoint pathway promotes cell survival in response to UV irradiation.Cancer Res. 2006 Mar 15;66(6):2997-3005. doi: 10.1158/0008-5472.CAN-05-3403. Cancer Res. 2006. PMID: 16540648 Free PMC article.
-
Xeroderma Pigmentosa Group A (XPA), Nucleotide Excision Repair and Regulation by ATR in Response to Ultraviolet Irradiation.Adv Exp Med Biol. 2017;996:41-54. doi: 10.1007/978-3-319-56017-5_4. Adv Exp Med Biol. 2017. PMID: 29124689 Free PMC article. Review.
-
Transcriptional and Posttranslational Regulation of Nucleotide Excision Repair: The Guardian of the Genome against Ultraviolet Radiation.Int J Mol Sci. 2016 Nov 4;17(11):1840. doi: 10.3390/ijms17111840. Int J Mol Sci. 2016. PMID: 27827925 Free PMC article. Review.
Cited by
-
Participation of keratinocyte- and fibroblast-derived factors in melanocyte homeostasis, the response to UV, and pigmentary disorders.Pigment Cell Melanoma Res. 2021 Jul;34(4):762-776. doi: 10.1111/pcmr.12985. Epub 2021 May 24. Pigment Cell Melanoma Res. 2021. PMID: 33973367 Free PMC article. Review.
-
The XPA Protein-Life under Precise Control.Cells. 2022 Nov 22;11(23):3723. doi: 10.3390/cells11233723. Cells. 2022. PMID: 36496984 Free PMC article. Review.
-
Sirtuin 1-mediated deacetylation of XPA DNA repair protein enhances its interaction with ATR protein and promotes cAMP-induced DNA repair of UV damage.J Biol Chem. 2018 Dec 7;293(49):19025-19037. doi: 10.1074/jbc.RA118.003940. Epub 2018 Oct 16. J Biol Chem. 2018. Retraction in: J Biol Chem. 2021 Jan-Jun;296:100185. doi: 10.1016/j.jbc.2020.100185. PMID: 30327428 Free PMC article. Retracted.
-
Progerin sequestration of PCNA promotes replication fork collapse and mislocalization of XPA in laminopathy-related progeroid syndromes.FASEB J. 2017 Sep;31(9):3882-3893. doi: 10.1096/fj.201700014R. Epub 2017 May 17. FASEB J. 2017. PMID: 28515154 Free PMC article.
-
Dietary proanthocyanidins inhibit UV radiation-induced skin tumor development through functional activation of the immune system.Mol Nutr Food Res. 2016 Jun;60(6):1374-82. doi: 10.1002/mnfr.201501026. Epub 2016 Apr 13. Mol Nutr Food Res. 2016. PMID: 26991736 Free PMC article. Review.
References
-
- Branzei D, Foiani M. Regulation of DNA repair throughout the cell cycle. Nat Rev Mol Cell Biol. 2008;9:297–308. - PubMed
-
- Sancar A, Lindsey-Boltz LA, Unsal-Kacmaz K, Linn S. Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu Rev Biochem. 2004;73:39–85. - PubMed
-
- Kraemer KH, Lee MM, Andrews AD, Lambert WC. The role of sunlight and DNA repair in melanoma and nonmelanoma skin cancer. The xeroderma pigmentosum paradigm. Arch Dermatol. 1994;130:1018–1021. - PubMed
-
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241–250. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

