Menstrual blood as a potential source of endometrial derived CD3+ T cells
- PMID: 22174921
- PMCID: PMC3235171
- DOI: 10.1371/journal.pone.0028894
Menstrual blood as a potential source of endometrial derived CD3+ T cells
Abstract
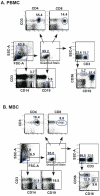
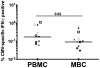
Studies of T cell-mediated immunity in the human female genital tract have been problematic due to difficulties associated with the collection of mucosal samples. Consequently, most studies rely on biopsies from the lower female genital tract or remnant tissue from hysterectomies. Availability of samples from healthy women is limited, as most studies are carried out in women with underlying pathologies. Menstruation is the cyclical sloughing off of endometrial tissue, and thus it should be a source of endometrial cells without the need for a biopsy. We isolated and phenotyped T cells from menstrual and peripheral blood and from endometrial biopsy-derived tissue from healthy women to determine the types of T cells present in this compartment. Our data demonstrated that T cells isolated from menstrual blood are a heterogeneous population of cells with markers reminiscent of blood and mucosal cells as well as unique phenotypes not represented in either compartment. T cells isolated from menstrual blood expressed increased levels of HLA-DR, αEβ7 and CXCR4 and reduced levels of CD62L relative to peripheral blood. Menstrual blood CD4+ T cells were enriched for cells expressing both CCR7 and CD45RA, markers identifying naïve T cells and were functional as determined by antigen-specific intracellular cytokine production assays. These data may open new avenues of investigation for cell mediated immune studies involving the female reproductive tract without the need for biopsies.
Conflict of interest statement
Figures
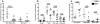


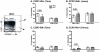

Similar articles
-
Menstrual blood closely resembles the uterine immune micro-environment and is clearly distinct from peripheral blood.Hum Reprod. 2014 Feb;29(2):303-14. doi: 10.1093/humrep/det398. Epub 2013 Nov 17. Hum Reprod. 2014. PMID: 24249743
-
Localization of T cells, interferon-gamma and HLA-DR in eutopic and ectopic human endometrium.Gynecol Obstet Invest. 1997;43(4):245-50. doi: 10.1159/000291866. Gynecol Obstet Invest. 1997. PMID: 9194623
-
Increased expressions of CD69 and HLA-DR but not of CD25 or CD71 on endometrial T lymphocytes of nonpregnant women.Hum Immunol. 1995 Mar;42(3):227-32. doi: 10.1016/0198-8859(94)00105-y. Hum Immunol. 1995. PMID: 7759310
-
Plasticity of human menstrual blood stem cells derived from the endometrium.J Zhejiang Univ Sci B. 2011 May;12(5):372-80. doi: 10.1631/jzus.B1100015. J Zhejiang Univ Sci B. 2011. PMID: 21528491 Free PMC article. Review.
-
Atypical Human Effector/Memory CD4+ T Cells With a Naive-Like Phenotype.Front Immunol. 2018 Dec 3;9:2832. doi: 10.3389/fimmu.2018.02832. eCollection 2018. Front Immunol. 2018. PMID: 30559746 Free PMC article. Review.
Cited by
-
Optimizing viable leukocyte sampling from the female genital tract for clinical trials: an international multi-site study.PLoS One. 2014 Jan 15;9(1):e85675. doi: 10.1371/journal.pone.0085675. eCollection 2014. PLoS One. 2014. PMID: 24454917 Free PMC article.
-
Is Neonatal Uterine Bleeding Involved in Early-Onset Endometriosis?Biomolecules. 2024 May 3;14(5):549. doi: 10.3390/biom14050549. Biomolecules. 2024. PMID: 38785956 Free PMC article. Review.
-
Human Tissue-Resident Memory T Cells in the Maternal-Fetal Interface. Lost Soldiers or Special Forces?Cells. 2020 Dec 16;9(12):2699. doi: 10.3390/cells9122699. Cells. 2020. PMID: 33339211 Free PMC article. Review.
-
The role of CD8+ T cells in endometriosis: a systematic review.Front Immunol. 2023 Jul 11;14:1225639. doi: 10.3389/fimmu.2023.1225639. eCollection 2023. Front Immunol. 2023. PMID: 37497226 Free PMC article.
-
Trophoblasts promote induction of a regulatory phenotype in B cells that can protect against detrimental T cell-mediated inflammation.Am J Reprod Immunol. 2019 Dec;82(6):e13187. doi: 10.1111/aji.13187. Epub 2019 Oct 6. Am J Reprod Immunol. 2019. PMID: 31487409 Free PMC article.
References
-
- Bulmer JN, Hagin SV, Browne CM, Billington WD. Localization of immunoglobulin-containing cells in human endometrium in the first trimester of pregnancy and throughout the menstrual cycle. Eur J Obstet Gynecol Reprod Biol. 1986;23:31–44. - PubMed
-
- Kelly JK, Fox H. The local immunological defence system of the human endometrium. J Reprod Immunol. 1979;1:39–45. - PubMed
-
- Russell MW, Mestecky J. Tolerance and protection against infection in the genital tract. Immunol Invest. 2010;39:500–525. - PubMed
-
- Yeaman GR, Guyre PM, Fanger MW, Collins JE, White HD, et al. Unique CD8+ T cell-rich lymphoid aggregates in human uterine endometrium. J Leukoc Biol. 1997;61:427–435. - PubMed
-
- Givan AL, White HD, Stern JE, Colby E, Gosselin EJ, et al. Flow cytometric analysis of leukocytes in the human female reproductive tract: comparison of fallopian tube, uterus, cervix, and vagina. Am J Reprod Immunol. 1997;38:350–359. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

