Molecular conversion of muscarinic acetylcholine receptor M(5) to muscarinic toxin 7 (MT7)-binding protein
- PMID: 22174976
- PMCID: PMC3237002
- DOI: 10.3390/toxins3111393
Molecular conversion of muscarinic acetylcholine receptor M(5) to muscarinic toxin 7 (MT7)-binding protein
Abstract
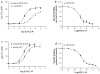
Muscarinic toxin 7 (MT7) is a mamba venom peptide that binds selectively to the M(1) muscarinic acetylcholine receptor. We have previously shown that the second (ECL2) and third (ECL3) extracellular loops of the M(1) receptor are critically involved in binding the peptide. In this study we used a mutagenesis approach on the M(5) subtype of the receptor family to find out if this possesses a similar structural architecture in terms of toxin binding as the M(1) receptor. An M(5) receptor construct (M(5)-E(175)Y(184)E(474)), mutated at the formerly deciphered critical residues on ECL2 and 3, gained the ability to bind MT7, but with rather low affinity as determined in a functional assay (apparent K(i) = 24 nM; apparent K(i) for M(1) = 0.5 nM). After screening for different domains and residues, we found a specific residue (P(179) to L in M(5)) in the middle portion of ECL2 that was necessary for high affinity binding of MT7 (M(5)-EL(179)YE, apparent K(i) = 0.5 nM). Mutation of P(179) to A confirmed a role for the leucine side chain in the binding of MT7. Together the results reveal new binding interactions between receptors and the MT7 peptide and strengthen the hypothesis that ECL2 sequence is of utmost importance for MT binding to muscarinic receptors.
Keywords: G protein-coupled receptor; acetylcholine receptor; ligand binding; muscarinic toxin.
Figures


References
-
- Caulfield M.P., Birdsall N.J. International Union of Pharmacology. XVII. Classification of muscarinic acetylcholine receptors. Pharmacol. Rev. 1998;50:279–290. - PubMed
-
- Wess J. Muscarinic acetylcholine receptor knockout mice: Novel phenotypes and clinical implications. Annu. Rev. Pharmacol. Toxicol. 2004;44:423–450. - PubMed
-
- Eglen R.M., Choppin A., Dillon M.P., Hegde S. Muscarinic receptor ligands and their therapeutic potential. Curr. Opin. Chem. Biol. 1999;3:426–432. - PubMed
-
- Eglen R.M., Choppin A., Watson N. Therapeutic opportunities from muscarinic receptor research. Trends Pharmacol. Sci. 2001;22:409–414. - PubMed
-
- Heinrich J.N., Butera J.A., Carrick T., Kramer A., Kowal D., Lock T., Marquis K.L., Pausch M.H., Popiolek M., Sun S.C., Tseng E., Uveges A.J., Mayer S.C. Pharmacological comparison of muscarinic ligands: Historical versus more recent muscarinic M1-preferring receptor agonists. Eur. J. Pharmacol. 2009;605:53–56. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

