Relationship between minority nonnucleoside reverse transcriptase inhibitor resistance mutations, adherence, and the risk of virologic failure
- PMID: 22179227
- PMCID: PMC3437231
- DOI: 10.1097/QAD.0b013e32834e9d7d
Relationship between minority nonnucleoside reverse transcriptase inhibitor resistance mutations, adherence, and the risk of virologic failure
Abstract
Objectives: To evaluate the risk of virologic failure conferred by suboptimal adherence to nonnucleoside reverse transcriptase inhibitors (NNRTIs) and minority NNRTI resistance mutations.
Design: Pooled analysis of the risk of virologic failure conferred by minority NNRTI resistance mutations and NNRTI adherence from three studies of treatment-naïve individuals initiating an NNRTI-based regimen.
Methods: Participants from each study were categorized into both adherence quartiles (Q1-Q4) and four strata: at least 95%, 80-94%, 60-79%, and below 60%. Weighted Cox proportional hazard models were used to estimate the risk of virologic failure.
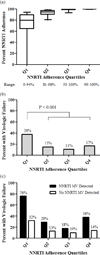
Results: The majority of participants (N = 768) had high measured adherence, but those in the lowest adherence quartile had the highest proportion of participants with virologic failure and the risk of virologic failure increased step-wise with adherence below 95%. Detection of minority NNRTI drug resistance mutations increased the proportion of participants with virologic failure across adherence quartiles (Cochran-Mantel-Haenszel P < 0.001) and adherence strata [Cochran-Mantel-Haenszel P < 0.001; <60% adherence, hazard ratio 1.7 (1.1-2.7), P = 0.02; 60-79% adherence, hazard ratio 1.2 (0.5-3.2), P = 0.67; 80-94% adherence, hazard ratio 2.5 (0.98-6.3), P = 0.06; ≥95% adherence, hazard ratio 3.6 (2.3-5.6), P < 0.001]. On multivariate analysis, the effect of minority variants was also most prominent at higher levels of medication adherence.
Conclusions: The presence of minority NNRTI resistance mutations and NNRTI adherence were found to be independent predictors of virologic failure, but also modify each other's effects on virologic failure. In addition to the focus on medication adherence counseling, ultrasensitive HIV-1 drug resistance assays could play a role in optimizing the success rates of first-line antiretroviral therapy.
Conflict of interest statement
Figures


References
-
- Bertagnolio S, Kelley K, Hassani AS, Obeng-Aduasare Y, Jordan M. Surveillance of transmitted and acquired HIV drug resistance using WHO surveys in resource-limited settings. 18th Conference on Retroviruses and Opportunistic Infections; February 27–March 2, 2011; Boston, MA.
-
- Gulick RM, Ribaudo HJ, Shikuma CM, Lustgarten S, Squires KE, Meyer WA, 3rd, et al. Triple-nucleoside regimens versus efavirenz-containing regimens for the initial treatment of HIV-1 infection. N Engl J Med. 2004;350:1850–1861. - PubMed
-
- Gulick RM, Ribaudo HJ, Shikuma CM, Lalama C, Schackman BR, Meyer WA, 3rd, et al. Three- vs four-drug antiretroviral regimens for the initial treatment of HIV-1 infection: a randomized controlled trial. JAMA. 2006;296:769–781. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

