Hcp family proteins secreted via the type VI secretion system coordinately regulate Escherichia coli K1 interaction with human brain microvascular endothelial cells
- PMID: 22184413
- PMCID: PMC3294675
- DOI: 10.1128/IAI.05994-11
Hcp family proteins secreted via the type VI secretion system coordinately regulate Escherichia coli K1 interaction with human brain microvascular endothelial cells
Abstract
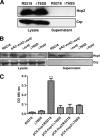
Type VI secretion systems (T6SSs) are involved in the pathogenicity of several gram-negative bacteria. Based on sequence analysis, we found that a cluster of Escherichia coli virulence factors (EVF) encoding a putative T6SS exists in the genome of the meningitis-causing E. coli K1 strain RS218. The T6SS-associated deletion mutants exhibited significant defects in binding to and invasion of human brain microvascular endothelial cells (HBMEC) compared with the parent strain. Hcp family proteins (the hallmark of T6SS), including Hcp1 and Hcp2, were localized in the bacterial outer membrane, but the involvements of Hcp1 and Hcp2 have been shown to differ in E. coli-HBMEC interaction. The deletion mutant of hcp2 showed defects in the bacterial binding to and invasion of HBMEC, while Hcp1 was secreted in a T6SS-dependent manner and induced actin cytoskeleton rearrangement, apoptosis, and the release of interleukin-6 (IL-6) and IL-8 in HBMEC. These findings demonstrate that the T6SS is functional in E. coli K1, and two Hcp family proteins participate in different steps of E. coli interaction with HBMEC in a coordinate manner, e.g., binding to and invasion of HBMEC, the cytokine and chemokine release followed by cytoskeleton rearrangement, and apoptosis in HBMEC. This is the first demonstration of the role of T6SS in meningitis-causing E. coli K1, and T6SS-associated Hcp family proteins are likely to contribute to the pathogenesis of E. coli meningitis.
Figures




Similar articles
-
The different roles of hcp1 and hcp2 of the type VI secretion system in Escherichia coli strain CE129.J Basic Microbiol. 2018 Nov;58(11):938-946. doi: 10.1002/jobm.201800156. Epub 2018 Sep 3. J Basic Microbiol. 2018. PMID: 30247772
-
YgfZ contributes to secretion of cytotoxic necrotizing factor 1 into outer-membrane vesicles in Escherichia coli.Microbiology (Reading). 2012 Mar;158(Pt 3):612-621. doi: 10.1099/mic.0.054122-0. Epub 2011 Dec 15. Microbiology (Reading). 2012. PMID: 22174383 Free PMC article.
-
Escherichia coli K1 RS218 interacts with human brain microvascular endothelial cells via type 1 fimbria bacteria in the fimbriated state.Infect Immun. 2005 May;73(5):2923-31. doi: 10.1128/IAI.73.5.2923-2931.2005. Infect Immun. 2005. PMID: 15845498 Free PMC article.
-
Current concepts on Escherichia coli K1 translocation of the blood-brain barrier.FEMS Immunol Med Microbiol. 2004 Nov 1;42(3):271-9. doi: 10.1016/j.femsim.2004.09.001. FEMS Immunol Med Microbiol. 2004. PMID: 15477040 Review.
-
Invasion processes of pathogenic Escherichia coli.Int J Med Microbiol. 2005 Oct;295(6-7):463-70. doi: 10.1016/j.ijmm.2005.07.004. Int J Med Microbiol. 2005. PMID: 16238020 Review.
Cited by
-
The Type VI Secretion System Modulates Flagellar Gene Expression and Secretion in Citrobacter freundii and Contributes to Adhesion and Cytotoxicity to Host Cells.Infect Immun. 2015 Jul;83(7):2596-604. doi: 10.1128/IAI.03071-14. Epub 2015 Apr 13. Infect Immun. 2015. PMID: 25870231 Free PMC article.
-
Blood-Brain Barrier Integrity Damage in Bacterial Meningitis: The Underlying Link, Mechanisms, and Therapeutic Targets.Int J Mol Sci. 2023 Feb 2;24(3):2852. doi: 10.3390/ijms24032852. Int J Mol Sci. 2023. PMID: 36769171 Free PMC article. Review.
-
The Missing Pieces: The Role of Secretion Systems in Campylobacter jejuni Virulence.Biomolecules. 2023 Jan 9;13(1):135. doi: 10.3390/biom13010135. Biomolecules. 2023. PMID: 36671522 Free PMC article. Review.
-
Genome analysis and in vivo virulence of porcine extraintestinal pathogenic Escherichia coli strain PCN033.BMC Genomics. 2015 Sep 21;16(1):717. doi: 10.1186/s12864-015-1890-9. BMC Genomics. 2015. PMID: 26391348 Free PMC article.
-
Designing a novel chimeric multi-epitope vaccine against Burkholderia pseudomallei, a causative agent of melioidosis.Front Med (Lausanne). 2022 Oct 18;9:945938. doi: 10.3389/fmed.2022.945938. eCollection 2022. Front Med (Lausanne). 2022. PMID: 36330071 Free PMC article.
References
-
- Aschtgen MS, Gavioli M, Dessen A, Lloubes R, Cascales E. 2010. The SciZ protein anchors the enteroaggregative Escherichia coli type VI secretion system to the cell wall. Mol. Microbiol. 75:886–899 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

