Competition and resilience between founder and introduced bacteria in the Caenorhabditis elegans gut
- PMID: 22184417
- PMCID: PMC3294642
- DOI: 10.1128/IAI.05522-11
Competition and resilience between founder and introduced bacteria in the Caenorhabditis elegans gut
Abstract
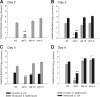
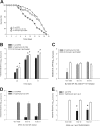
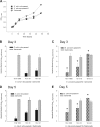
The microbial communities that reside within the intestinal tract in vertebrates are complex and dynamic. In this report, we establish the utility of Caenorhabditis elegans as a model system for identifying the factors that contribute to bacterial persistence and for host control of gut luminal populations. We found that for N2 worms grown on mixed lawns of bacteria, Salmonella enterica serovar Typhimurium substantially outcompeted Escherichia coli, even when E. coli was initially present at 100-fold-higher concentrations. To address whether innate immunity affects the competition, the daf-2 and daf-16 mutants were studied; their total gut bacterial levels reflect overall capacity for colonization, but Salmonella outcompeted E. coli to an extent similar to wild-type worms. To address the role of virulence properties, Salmonella Δspi-1 Δspi-2 was used to compete with E. coli. The net differential was significantly less than that for wild-type Salmonella; thus, spi-1 spi-2 encodes C. elegans colonization factors. An E. coli strain with repeated in vivo passage had an enhanced ability to compete against an in vitro-passed E. coli strain and against Salmonella. Our data provide evidence of active competition for colonization niches in the C. elegans gut, as determined by bacterial factors and subject to in vivo selection.
Figures


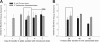



Similar articles
-
The two-component sensor kinase KdpD is required for Salmonella typhimurium colonization of Caenorhabditis elegans and survival in macrophages.Cell Microbiol. 2011 Oct;13(10):1618-37. doi: 10.1111/j.1462-5822.2011.01645.x. Epub 2011 Aug 24. Cell Microbiol. 2011. PMID: 21790938
-
Salmonella Typhimurium utilizes a T6SS-mediated antibacterial weapon to establish in the host gut.Proc Natl Acad Sci U S A. 2016 Aug 23;113(34):E5044-51. doi: 10.1073/pnas.1608858113. Epub 2016 Aug 8. Proc Natl Acad Sci U S A. 2016. PMID: 27503894 Free PMC article.
-
Resistance to antimicrobial peptides contributes to persistence of Salmonella typhimurium in the C. elegans intestine.Cell Microbiol. 2008 Jun;10(6):1259-73. doi: 10.1111/j.1462-5822.2008.01124.x. Epub 2008 Jan 21. Cell Microbiol. 2008. PMID: 18221392
-
Identification of virulence properties in Salmonella Typhimurium DT104 using Caenorhabditis elegans.PLoS One. 2013 Oct 4;8(10):e76673. doi: 10.1371/journal.pone.0076673. eCollection 2013. PLoS One. 2013. PMID: 24124587 Free PMC article.
-
Deletion of Invasion Protein B in Salmonella enterica Serovar Typhimurium Influences Bacterial Invasion and Virulence.Curr Microbiol. 2015 Dec;71(6):687-92. doi: 10.1007/s00284-015-0903-x. Epub 2015 Sep 4. Curr Microbiol. 2015. PMID: 26341924
Cited by
-
Modeling Host-Microbiome Interactions in Caenorhabditis elegans.J Nematol. 2017 Dec;49(4):348-356. J Nematol. 2017. PMID: 29353922 Free PMC article.
-
Biotic interactions between the human pathogen Legionella pneumophila and nematode grazers in cooling tower biofilms.PLoS One. 2024 Oct 25;19(10):e0309820. doi: 10.1371/journal.pone.0309820. eCollection 2024. PLoS One. 2024. PMID: 39453963 Free PMC article.
-
Association with a novel protective microbe facilitates host adaptation to a stressful environment.Evol Lett. 2021 Mar 17;5(2):118-129. doi: 10.1002/evl3.223. eCollection 2021 Apr. Evol Lett. 2021. PMID: 33868708 Free PMC article.
-
Host Immunity Alters Community Ecology and Stability of the Microbiome in a Caenorhabditis elegans Model.mSystems. 2021 Apr 20;6(2):e00608-20. doi: 10.1128/mSystems.00608-20. mSystems. 2021. PMID: 33879498 Free PMC article.
-
Immune-mediated competition benefits protective microbes over pathogens in a novel host species.Heredity (Edinb). 2022 Dec;129(6):327-335. doi: 10.1038/s41437-022-00569-3. Epub 2022 Nov 9. Heredity (Edinb). 2022. PMID: 36352206 Free PMC article.
References
-
- Aballay A, Ausubel FM. 2002. Caenorhabditis elegans as a host for the study of host-pathogen interactions. Curr. Opin. Microbiol. 5:97–101 - PubMed
-
- Aballay A, Drenkard E, Hilbun LR, Ausubel FM. 2003. Caenorhabditis elegans innate immune response triggered by Salmonella enterica requires intact LPS and is mediated by a MAPK signaling pathway. Curr. Biol. 13:47–52 - PubMed
-
- Aballay A, Yorgey P, Ausubel FM. 2000. Salmonella typhimurium proliferates and establishes a persistent infection in the intestine of Caenorhabditis elegans. Curr. Biol. 10:1539–1542 - PubMed
-
- Alegado RA, Tan MW. 2008. Resistance to antimicrobial peptides contributes to persistence of Salmonella typhimurium in the C. elegans intestine. Cell. Microbiol. 10:1259–1273 - PubMed
-
- Apfeld J, Kenyon C. 1999. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402:804–809 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

