Regulating the regulator: post-translational modification of RAS
- PMID: 22189424
- PMCID: PMC3879958
- DOI: 10.1038/nrm3255
Regulating the regulator: post-translational modification of RAS
Abstract
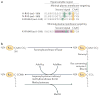
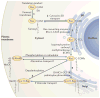
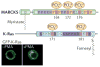
RAS proteins are monomeric GTPases that act as binary molecular switches to regulate a wide range of cellular processes. The exchange of GTP for GDP on RAS is regulated by guanine nucleotide exchange factors (GEFs) and GTPase-activating proteins (GAPs), which regulate the activation state of RAS without covalently modifying it. By contrast, post-translational modifications (PTMs) of RAS proteins direct them to various cellular membranes and, in some cases, modulate GTP-GDP exchange. Important RAS PTMs include the constitutive and irreversible remodelling of its carboxy-terminal CAAX motif by farnesylation, proteolysis and methylation, reversible palmitoylation, and conditional modifications, including phosphorylation, peptidyl-prolyl isomerisation, monoubiquitylation, diubiquitylation, nitrosylation, ADP ribosylation and glucosylation.
Figures



References
-
- Malumbres M, Barbacid M. RAS oncogenes: the first 30 years. Nat Rev Cancer. 2003;3:459–65. - PubMed
-
- Parada LF, Tabin CJ, Shih C, Weinberg RA. Human EJ bladder carcinoma oncogene is homologue of Harvey sarcoma virus ras gene. Nature. 1982;297:474–8. - PubMed
-
- Santos E, Tronick SR, Aaronson SA, Pulciani S, Barbacid M. T24 human bladder carcinoma oncogene is an activated form of the normal human homologue of BALB- and Harvey-MSV transforming genes. Nature. 1982;298:343–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

