Mechanisms underlying reductant-induced reactive oxygen species formation by anticancer copper(II) compounds
- PMID: 22189939
- PMCID: PMC3371635
- DOI: 10.1007/s00775-011-0864-x
Mechanisms underlying reductant-induced reactive oxygen species formation by anticancer copper(II) compounds
Abstract
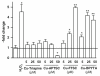
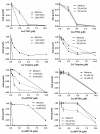
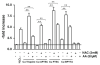
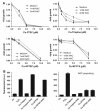
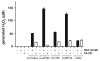
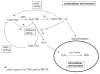
Intracellular generation of reactive oxygen species (ROS) via thiol-mediated reduction of copper(II) to copper(I) has been assumed as the major mechanism underlying the anticancer activity of copper(II) complexes. The aim of this study was to compare the anticancer potential of copper(II) complexes of Triapine (3-aminopyridine-2-carboxaldehyde thiosemicarbazone; currently in phase II clinical trials) and its terminally dimethylated derivative with that of 2-formylpyridine thiosemicarbazone and that of 2,2'-bipyridyl-6-carbothioamide. Experiments on generation of oxidative stress and the influence of biologically relevant reductants (glutathione, ascorbic acid) on the anticancer activity of the copper complexes revealed that reductant-dependent redox cycling occurred mainly outside the cells, leading to generation and dismutation of superoxide radicals resulting in cytotoxic amounts of H(2)O(2). However, without extracellular reductants only weak intracellular ROS generation was observed at IC(50) levels, suggesting that cellular thiols are not involved in copper-complex-induced oxidative stress. Taken together, thiol-induced intracellular ROS generation might contribute to the anticancer activity of copper thiosemicarbazone complexes but is not the determining factor.
Figures






Similar articles
-
Ribonucleotide reductase inhibition by metal complexes of Triapine (3-aminopyridine-2-carboxaldehyde thiosemicarbazone): a combined experimental and theoretical study.J Inorg Biochem. 2011 Nov;105(11):1422-31. doi: 10.1016/j.jinorgbio.2011.07.003. Epub 2011 Jul 31. J Inorg Biochem. 2011. PMID: 21955844 Free PMC article.
-
Increased generation of intracellular reactive oxygen species initiates selective cytotoxicity against the MCF-7 cell line resultant from redox active combination therapy using copper-thiosemicarbazone complexes.J Biol Inorg Chem. 2016 Jun;21(3):407-19. doi: 10.1007/s00775-016-1350-2. Epub 2016 Mar 7. J Biol Inorg Chem. 2016. PMID: 26951232
-
Strong effect of copper(II) coordination on antiproliferative activity of thiosemicarbazone-piperazine and thiosemicarbazone-morpholine hybrids.Dalton Trans. 2015 May 21;44(19):9071-90. doi: 10.1039/c5dt01076d. Dalton Trans. 2015. PMID: 25896351
-
High Copper Complex Stability and Slow Reduction Kinetics as Key Parameters for Improved Activity, Paraptosis Induction, and Impact on Drug-Resistant Cells of Anticancer Thiosemicarbazones.Antioxid Redox Signal. 2020 Aug 20;33(6):395-414. doi: 10.1089/ars.2019.7854. Epub 2020 Jun 9. Antioxid Redox Signal. 2020. PMID: 32336116
-
Recent Advances in Copper Intercalators as Anticancer Agents.J Fluoresc. 2018 Sep;28(5):1195-1205. doi: 10.1007/s10895-018-2283-7. Epub 2018 Aug 31. J Fluoresc. 2018. PMID: 30171479 Review.
Cited by
-
Major vault protein supports glioblastoma survival and migration by upregulating the EGFR/PI3K signalling axis.Oncotarget. 2013 Nov;4(11):1904-18. doi: 10.18632/oncotarget.1264. Oncotarget. 2013. PMID: 24243798 Free PMC article.
-
Binding Models of Copper(II) Thiosemicarbazone Complexes with Human Serum Albumin: A Speciation Study.Molecules. 2021 May 5;26(9):2711. doi: 10.3390/molecules26092711. Molecules. 2021. PMID: 34063080 Free PMC article.
-
Liposomal formulations of anticancer copper(II) thiosemicarbazone complexes.Dalton Trans. 2021 Nov 16;50(44):16053-16066. doi: 10.1039/d1dt02763h. Dalton Trans. 2021. PMID: 34617075 Free PMC article.
-
β-Carboline copper complex as a potential mitochondrial-targeted anticancer chemotherapeutic agent: Favorable attenuation of human breast cancer MCF7 cells via apoptosis.Saudi J Biol Sci. 2020 Aug;27(8):2164-2173. doi: 10.1016/j.sjbs.2020.05.001. Epub 2020 May 8. Saudi J Biol Sci. 2020. PMID: 32714043 Free PMC article.
-
Destruxins: fungal-derived cyclohexadepsipeptides with multifaceted anticancer and antiangiogenic activities.Biochem Pharmacol. 2013 Aug 1;86(3):361-77. doi: 10.1016/j.bcp.2013.05.022. Epub 2013 Jun 6. Biochem Pharmacol. 2013. PMID: 23747344 Free PMC article.
References
-
- Gupte A, Mumper RJ. Cancer Treat Rev. 2009;35:32–46. - PubMed
-
- Trachootham D, Alexandre J, Huang P. Nat Rev Drug Discov. 2009;8:579–591. - PubMed
-
- Denny WA. Future Oncol. 2010;6:419–428. doi:10.2217/fon.10.1. - PubMed
-
- Hartinger CG, Zorbas-Seifried S, Jakupec MA, Kynast B, Zorbas H, Keppler BK. J Inorg Biochem. 2006;100:891–904. - PubMed
-
- Hall MD, Hambley TW. Coord Chem Rev. 2002;232:49–67.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

