Nerve growth factor is associated with islet graft failure following intraportal transplantation
- PMID: 22192949
- PMCID: PMC3365801
- DOI: 10.4161/isl.18467
Nerve growth factor is associated with islet graft failure following intraportal transplantation
Abstract
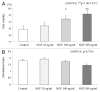
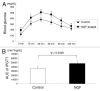
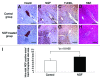
Nerve growth factor (NGF) has recently been recognized as an angiogenic factor with an important regulatory role in pancreatic β-cell function. We previously showed that treatment of pancreatic islets with NGF improved their quality and viability. Revascularization and survival of islets transplanted under the kidney capsule were improved by NGF. However, the usefulness of NGF in intraportal islet transplantation was not previously tested. To resolve this problem, we transplanted syngeneic islets (360 islet equivalents per recipient) cultured with or without NGF into the portal vein of streptozotocin-induced diabetic BALB/c mice. Analysis revealed that 44.4% (4/9) of control and 12.5% (1/8) of NGF-treated mice attained normoglycemia (≤ 200 mg/dL) (p = 0.195). NGF-treated islets led to worse graft function (area under the curve of intraperitoneal glucose tolerance tests (IPGTT) on post-operative day (POD) 30, control; 35,800 ± 3,960 min*mg/dl, NGF-treated; 47,900 ± 3,220 min*mg/dl: *p = 0.0348). NGF treatment of islets was also associated with increased graft failure [the percentage of TdT-mediated dUTP-biotin nick-end labeling (TUNEL)-positive and necrotic transplanted islets on POD 5, control; 23.8% (5/21), NGF-treated; 52.9% (9/17): p = 0.0650] following intraportal islet transplantation. Nonviable (TUNEL-positive and necrotic) islets in both groups expressed vascular endothelial growth factor (VEGF) and hypoxia-inducible factor-1α (HIF-1α). On the other hand, viable (TUNEL-negative and not necrotic) islets in both groups did not express VEGF and HIF-1α. In the present study, pre-transplant NGF treatment was associated with impaired survival and angiogenesis of intraportal islet grafts. The effect of NGF on islet transplantation may significantly vary according to the transplant site.
Keywords: apoptosis; diabetes; intraportal; islet transplantation; nerve growth factor.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
