Safety and immunogenicity profile of human papillomavirus-16/18 AS04 adjuvant cervical cancer vaccine: a randomized controlled trial in healthy adolescent girls of Bangladesh
- PMID: 22194637
- PMCID: PMC3244935
- DOI: 10.1093/jjco/hyr173
Safety and immunogenicity profile of human papillomavirus-16/18 AS04 adjuvant cervical cancer vaccine: a randomized controlled trial in healthy adolescent girls of Bangladesh
Abstract
Aim: Bangladesh has the highest level of incidence and mortality rates due to cervical cancer among women. The prevalence of cervical cancer in Bangladeshi women is 25-30/100,000. Human papillomavirus is an important cause of cervical cancer. The study was conducted to assess the immunogenicity and safety profile of human papillomavirus-16/18 AS04-adjuvanted cervical cancer vaccines in healthy Bangladeshi girls aged 9-13 years. Procedure This was a randomized (3:1) controlled trial with two parallel groups, the vaccine and control groups, that included 67 participants in Bangladesh. Subjects were given GlaxoSmithKline human papillomavirus-16/18 AS04-adjuvanted cervical cancer vaccine (and controls no vaccine) at the first day of vaccination (Day 0), at 1- and 6-month schedule and followed up until 7 months. Blood samples were taken for human papillomavirus antibody at enrollment and 1 month post-schedule at Month 7 from both subjects and controls. Safety data were gathered throughout the study period.
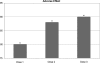
Results: Fifty subjects received vaccine at Day 0, 1 month and 6 months. All subjects were initially sero-negative in the vaccine group, and developed sero-conversion for human papillomavirus-16 and -18 antibodies except for one at Month 7. Seventeen controls did not receive vaccine. Clients were followed up for serious medically important events and blood samples were taken for human papillomavirus antibody detection at Day 0 and Month 7. Sero-conversion was found in 97.5% of subjects and no sero-conversion was found in the controls. Bivalent human papillomavirus vaccine was generally well tolerated, with no vaccine-related serious adverse experiences.
Conclusions: The human papillomavirus-16/18 AS04-adjuvanted vaccine was generally well tolerated and highly immunogenic when administered to young adolescent females and could be a promising tool for the prevention and control of cervical cancer in Bangladesh.
Figures
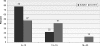
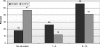
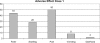


References
-
- Boyle P, Levin B. International Agency for Research on Cancer; 2008. World Cancer Report 2008; pp. 128–9.
-
- Globocan. International Agency for Research on Cancer; 2008. Bangladeshdata.iarc.factsheets.pdf .
-
- Dinshaw K, Mishra G, Shastri S. Determinants of compliance in a cluster randomised controlled trial on screening of breast and cervix cancer in Mumbai, India.1. Compliance to screening. Oncology. 2007;73:145–53. - PubMed
-
- Bosch FX, de Sanjose S. Chapter 1: human papilloma virus and cervical cancer—burden and assessment of causality. J Natl cancer Inst Monogr. 2003;1:3–13. - PubMed
-
- Ashrafunnessa, Khatun SS, Chowdhury TA, Shamsuddin L, Islam MN, Hassan MS, et al. Human papilloma virus in cervical intraepithelial neoplasia in Bangladesh. Bangladesh J Obstet Gynaecol. 2005;20:13–8.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

