The real-time-based assessment of the microbial killing by the antimicrobial compounds of neutrophils
- PMID: 22194669
- PMCID: PMC3236378
- DOI: 10.1100/2011/376278
The real-time-based assessment of the microbial killing by the antimicrobial compounds of neutrophils
Abstract
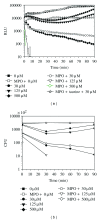
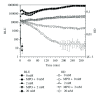
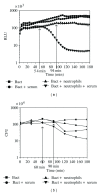
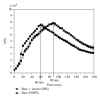
A recombinant Escherichia coli K-12 strain, transformed with a modified bacterial luciferase gene (luxABCDE) from Photorhabdus luminescens, was constructed in order to monitor the activity of various antimicrobial agents on a real-time basis. This E. coli-lux emitted, without any addition of substrate, constitutive bioluminescence (BL), which correlated to the number of viable bacterial cells. The decrease in BL signal correlated to the number of killed bacterial cells. Antimicrobial activity of hydrogen peroxide (H(2)O(2)) and myeloperoxidase (MPO) was assessed. In high concentrations, H(2)O(2) alone had a bacteriocidic function and MPO enhanced this killing by forming hypochlorous acid (HOCl). Taurine, the known HOCl scavenger, blocked the killing by MPO. When E. coli-lux was incubated with neutrophils, similar killing kinetics was recorded as in H(2)O(2)/MPO experiments. The opsonization of bacteria enhanced the killing, and the maximum rate of the MPO release from lysosomes coincided with the onset of the killing.
Keywords: acid; bacterial; bioluminescence; hydrogen; hypochlorous; luciferase; myeloperoxidase; neutrophil; peroxide; phagocytosis.
Figures
Similar articles
-
Escherichia coli K-12 (pEGFPluxABCDEamp): a tool for analysis of bacterial killing by antibacterial agents and human complement activities on a real-time basis.Luminescence. 2013 Sep-Oct;28(5):771-9. doi: 10.1002/bio.2435. Epub 2012 Nov 6. Luminescence. 2013. PMID: 23129448
-
A real-time-based in vitro assessment of the oxidative antimicrobial mechanisms of the myeloperoxidase-hydrogen peroxide-halide system.Mol Immunol. 2019 Dec;116:38-44. doi: 10.1016/j.molimm.2019.09.017. Epub 2019 Oct 5. Mol Immunol. 2019. PMID: 31593870
-
Neutrophil granule proteins generate bactericidal ammonia chloramine on reaction with hydrogen peroxide.Free Radic Biol Med. 2017 Dec;113:363-371. doi: 10.1016/j.freeradbiomed.2017.10.343. Epub 2017 Oct 18. Free Radic Biol Med. 2017. PMID: 29055823
-
Redox reactions and microbial killing in the neutrophil phagosome.Antioxid Redox Signal. 2013 Feb 20;18(6):642-60. doi: 10.1089/ars.2012.4827. Epub 2012 Oct 9. Antioxid Redox Signal. 2013. PMID: 22881869 Review.
-
Neutrophils as Sentinel Cells of the Immune System: A Role of the MPO-halide-system in Innate and Adaptive Immunity.Curr Med Chem. 2020;27(17):2840-2851. doi: 10.2174/0929867326666190819123300. Curr Med Chem. 2020. PMID: 31424363 Review.
Cited by
-
Role of oxidative stress in infectious diseases. A review.Folia Microbiol (Praha). 2013 Nov;58(6):503-13. doi: 10.1007/s12223-013-0239-5. Epub 2013 Mar 17. Folia Microbiol (Praha). 2013. PMID: 23504625 Review.
-
Interplay between enterobactin, myeloperoxidase and lipocalin 2 regulates E. coli survival in the inflamed gut.Nat Commun. 2015 May 12;6:7113. doi: 10.1038/ncomms8113. Nat Commun. 2015. PMID: 25964185 Free PMC article.
-
Epigallocatechin-3-Gallate Inhibition of Myeloperoxidase and Its Counter-Regulation by Dietary Iron and Lipocalin 2 in Murine Model of Gut Inflammation.Am J Pathol. 2016 Apr;186(4):912-26. doi: 10.1016/j.ajpath.2015.12.004. Epub 2016 Mar 8. Am J Pathol. 2016. PMID: 26968114 Free PMC article.
-
Investigating the short- and long-term effects of antibacterial agents using a real-time assay based on bioluminescent E. coli-lux.Heliyon. 2020 Jul 2;6(7):e04232. doi: 10.1016/j.heliyon.2020.e04232. eCollection 2020 Jul. Heliyon. 2020. PMID: 32642580 Free PMC article.
-
Mutant allele knockout with novel CRISPR nuclease promotes myelopoiesis in ELANE neutropenia.Mol Ther Methods Clin Dev. 2022 Jun 9;26:119-131. doi: 10.1016/j.omtm.2022.06.002. eCollection 2022 Sep 8. Mol Ther Methods Clin Dev. 2022. PMID: 35795780 Free PMC article.
References
-
- Anderson KL, Smith KA, Pio F, Torbett BE, Maki RA. Neutrophils deficient in PU.1 do not terminally differentiate or become functionally competent. Blood. 1998;92(5):1576–1585. - PubMed
-
- Lieber JG, Webb S, Suratt BT, et al. The in vitro production and characterization of neutrophils from embryonic stem cells. Blood. 2004;103(3):852–859. - PubMed
-
- Hayashi F, Means TK, Luster AD. Toll-like receptors stimulate human neutrophil function. Blood. 2003;102(7):2660–2669. - PubMed
-
- Cassatella MA, Locati M, Mantovani A. Never underestimate the power of a neutrophil. Immunity. 2009;31(5):698–700. - PubMed
-
- Dale DC, Boxer L, Liles WC. The phagocytes: neutrophils and monocytes. Blood. 2008;112(4):935–945. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

