Modeling Ca2+ signaling in the microcirculation: intercellular communication and vasoreactivity
- PMID: 22196162
- PMCID: PMC3681513
- DOI: 10.1615/critrevbiomedeng.v39.i5.50
Modeling Ca2+ signaling in the microcirculation: intercellular communication and vasoreactivity
Abstract
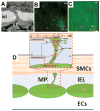
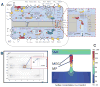
A network of intracellular signaling pathways and complex intercellular interactions regulate calcium mobilization in vascular cells, arteriolar tone, and blood flow. Different endothelium-derived vasoreactive factors have been identified and the importance of myoendothelial communication in vasoreactivity is now well appreciated. The ability of many vascular networks to conduct signals upstream also is established. This phenomenon is critical for both short-term changes in blood perfusion as well as long-term adaptations of a vascular network. In addition, in a phenomenon termed vasomotion, arterioles often exhibit spontaneous oscillations in diameter. This is thought to improve tissue oxygenation and enhance blood flow. Experimentation has begun to reveal important aspects of the regulatory machinery and the significance of these phenomena for the regulation of local perfusion and oxygenation. Mathematical modeling can assist in elucidating the complex signaling mechanisms that participate in these phenomena. This review highlights some of the important experimental studies and relevant mathematical models that provide the current understanding of these mechanisms in vasoreactivity.
Figures


References
-
- Sandow SL, Haddock RE, Hill CE, Chadha PS, Kerr PM, Welsh DG, et al. What’s where and why at a vascular myoendothelial microdomain signalling complex. Clin Exp Pharmacol Physiol. 2009;36(1):67–76. - PubMed
-
- Tallini YN, Brekke JF, Shui B, Doran R, Hwang SM, Nakai J, et al. Propagated endothelial Ca2+ waves and arteriolar dilation in vivo: measurements in Cx40BAC GCaMP2 transgenic mice. Circ Res. 2007;101(12):1300–9. - PubMed
-
- Luo CH, Rudy Y. A dynamic model of the cardiac ventricular action potential. I. Simulations of ionic currents and concentration changes. Circ Res. 1994;74(6):1071–96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

