The pore of the voltage-gated proton channel
- PMID: 22196334
- PMCID: PMC3244940
- DOI: 10.1016/j.neuron.2011.11.014
The pore of the voltage-gated proton channel
Abstract
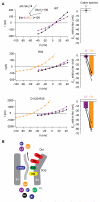
In classical tetrameric voltage-gated ion channels four voltage-sensing domains (VSDs), one from each subunit, control one ion permeation pathway formed by four pore domains. The human Hv1 proton channel has a different architecture, containing a VSD, but lacking a pore domain. Since its location is not known, we searched for the Hv permeation pathway. We find that mutation of the S4 segment's third arginine R211 (R3) compromises proton selectivity, enabling conduction of a metal cation and even of the large organic cation guanidinium, reminiscent of Shaker's omega pore. In the open state, R3 appears to interact with an aspartate (D112) that is situated in the middle of S1 and is unique to Hv channels. The double mutation of both residues further compromises cation selectivity. We propose that membrane depolarization reversibly positions R3 next to D112 in the transmembrane VSD to form the ion selectivity filter in the channel's open conformation.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures





References
-
- Baker OS, Larsson HP, Mannuzzu LM, Isacoff EY. Three transmembrane conformations and sequence-dependent displacement of the s4 domain in shaker k+ channel gating. Neuron. 1998;20(6):1283–1294. - PubMed
-
- DeCoursey TE. Voltage-gated proton channels and other proton transfer pathways. Physiol Rev. 2003;83(2):475–579. - PubMed
-
- DeCoursey TE, Morgan D, Cherny VV. The voltage dependence of nadph oxidase reveals why phagocytes need proton channels. Nature. 2003;422(6931):531–534. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

