Cefoperazone-treated mice as an experimental platform to assess differential virulence of Clostridium difficile strains
- PMID: 22198617
- PMCID: PMC3337121
- DOI: 10.4161/gmic.19142
Cefoperazone-treated mice as an experimental platform to assess differential virulence of Clostridium difficile strains
Abstract
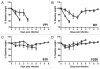
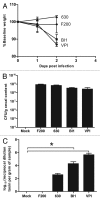
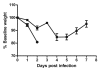
The toxin-producing bacterium C. difficile is the leading cause of antibiotic-associated colitis, with an estimated 500,000 cases C. difficile infection (CDI) each year in the US with a cost approaching 3 billion dollars. Despite the significance of CDI, the pathogenesis of this infection is still being defined. The recent development of tractable murine models of CDI will help define the determinants of C. difficile pathogenesis in vivo. To determine if cefoperazone-treated mice could be utilized to reveal differential pathogenicity of C. difficile strains, 5-8 week old C57BL/6 mice were pretreated with a 10 d course of cefoperazone administered in the drinking water. Following a 2-d recovery period without antibiotics, the animals were orally challenged with C. difficile strains chosen to represent the potential range of virulence of this organism from rapidly fatal to nonpathogenic. Animals were monitored for loss of weight and clinical signs of colitis. At the time of harvest, C. difficile strains were isolated from cecal contents and the severity of colitis was determined by histopathologic examination of the cecum and colon. Cefoperazone treated mice challenged with C. difficile strains VPI 10463 and BI1 exhibited signs of severe colitis while infection with 630 and F200 was subclinical. This increased clinical severity was correlated with more severe histopathology with significantly more edema, inflammation and epithelial damage encountered in the colons of animals infected with VPI 10463 and BI1. Disease severity also correlated with levels of C. difficile cytotoxic activity in intestinal tissues and elevated blood neutrophil counts. Cefoperazone treated mice represent a useful model of C. difficile infection that will help us better understand the pathogenesis and virulence of this re-emerging pathogen.
Figures
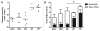

References
-
- Hall JC, O'Toole E. Intestinal flora in new-born infants with a description of a new pathogenic anaerobe, Bacillus difficilis. Am J Dis Child. 1935;49:390–402.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
