Apoptotic and anti-metastatic effects of the whole skin of Venenum bufonis in A549 human lung cancer cells
- PMID: 22200726
- PMCID: PMC3584623
- DOI: 10.3892/ijo.2011.1310
Apoptotic and anti-metastatic effects of the whole skin of Venenum bufonis in A549 human lung cancer cells
Abstract
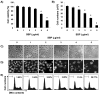
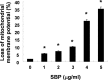
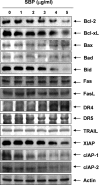
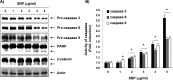
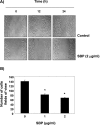
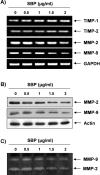
In the present study, the effects of the whole skin of Venenum bufonis on apoptotic and anti-invasive activity in A549 human lung cancer cells were investigated. Treatment with extract of the whole skin of V. bufonis (SVB) resulted in a significant decrease in cell growth of A549 cells, depending on dosage, which was associated with apoptosis induction, as proved by chromatin condensation and accumulation of apoptotic fraction. SVB treatment induced expression of death receptor-related proteins, such as death receptor 4, which further triggered activation of caspase-8 and cleavage of Bid. In addition, the increase in apoptosis by SVB treatment was correlated with dysfunction of mitochondria, activation of caspase-9 and -3, downregulation of IAP family proteins, such as XIAP, cIAP-1 and cIAP-2, and concomitant degradation of activated caspase-3-specific target proteins, such as poly (ADP-ribose) polymerase and β-catenin proteins. However, z-DEVD-fmk, a caspase-3-specific inhibitor, blocked SVB-induced apoptosis and increased the survival rate of SVB-treated cells, indicating that activation of caspase-3 plays a key role in SVB-induced apoptosis. In addition, within concentrations that were not cytotoxic to A549 cells, SVB induced marked inhibition of cell motility and invasiveness. Activities of matrix metalloproteinase (MMP)-2 and MMP-9 in AGS cells were dose-dependently inhibited by treatment with SVB, and this was also correlated with a decrease in expression of their mRNA and proteins, and upregulation of tissue inhibitors of metalloproteinase (TIMP)-1 and TIMP-2 mRNA expression. Further studies are needed; however, the results indicated that SVB induces apoptosis of A549 cells through a signaling cascade of death receptor-mediated extrinsic as well as mitochondria-mediated intrinsic caspase pathways. Our data also demonstrated that MMPs are critical targets of SVB-induced anti-invasiveness in A549 cells.
Figures

Similar articles
-
Apoptosis induction of human lung carcinoma cells by Chan Su (Venenum Bufonis) through activation of caspases.J Acupunct Meridian Stud. 2009 Sep;2(3):210-7. doi: 10.1016/S2005-2901(09)60057-1. J Acupunct Meridian Stud. 2009. PMID: 20633494
-
Casticin induces caspase-mediated apoptosis via activation of mitochondrial pathway and upregulation of DR5 in human lung cancer cells.Asian Pac J Trop Med. 2013 May 13;6(5):372-8. doi: 10.1016/S1995-7645(13)60041-3. Asian Pac J Trop Med. 2013. PMID: 23608376
-
Induction of apoptosis in human lung carcinoma cells by the water extract of Panax notoginseng is associated with the activation of caspase-3 through downregulation of Akt.Int J Oncol. 2009 Jul;35(1):121-7. doi: 10.3892/ijo_00000320. Int J Oncol. 2009. PMID: 19513559
-
A methyl jasmonate derivative, J-7, induces apoptosis in human hepatocarcinoma Hep3B cells in vitro.Toxicol In Vitro. 2010 Oct;24(7):1920-6. doi: 10.1016/j.tiv.2010.08.001. Epub 2010 Aug 7. Toxicol In Vitro. 2010. PMID: 20696234
-
Apoptotic signaling pathways in bone metastatic lung cancer: a comprehensive analysis.Discov Oncol. 2024 Jul 26;15(1):310. doi: 10.1007/s12672-024-01151-5. Discov Oncol. 2024. PMID: 39060849 Free PMC article. Review.
Cited by
-
Cinobufagin Induces Cell Cycle Arrest at the G2/M Phase and Promotes Apoptosis in Malignant Melanoma Cells.Front Oncol. 2019 Sep 4;9:853. doi: 10.3389/fonc.2019.00853. eCollection 2019. Front Oncol. 2019. PMID: 31552178 Free PMC article.
-
Boehmenan, a Lignan From the Chinese Medicinal Plant Clematis armandii, Inhibits A431 Cell Growth via Blocking p70S6/S6 Kinase Pathway.Integr Cancer Ther. 2017 Sep;16(3):351-359. doi: 10.1177/1534735416669803. Epub 2016 Oct 3. Integr Cancer Ther. 2017. PMID: 27698262 Free PMC article.
-
Fraxetin Inhibits the Proliferation and Metastasis of Glioma Cells by Inactivating JAK2/STAT3 Signaling.Evid Based Complement Alternat Med. 2021 Apr 16;2021:5540139. doi: 10.1155/2021/5540139. eCollection 2021. Evid Based Complement Alternat Med. 2021. PMID: 33959183 Free PMC article.
-
TIMP-1 Inhibits Apoptosis in Lung Adenocarcinoma Cells via Interaction with Bcl-2.PLoS One. 2015 Sep 14;10(9):e0137673. doi: 10.1371/journal.pone.0137673. eCollection 2015. PLoS One. 2015. PMID: 26366732 Free PMC article.
-
Effectiveness and Mechanism of Resibufogenin on Human Renal Cancer Cell Caki-1.Biology (Basel). 2024 Dec 19;13(12):1064. doi: 10.3390/biology13121064. Biology (Basel). 2024. PMID: 39765731 Free PMC article.
References
-
- Jin Z, El-Deiry WS. Overview of cell death signaling pathways. Cancer Biol Ther. 2005;4:139–163. - PubMed
-
- Earnshaw WC, Martins LM, Kaufmann SH. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu Rev Biochem. 1999;6:383–424. - PubMed
-
- Stennicke HR, Salvesen GS. Properties of the caspases. Biochim Biophys Acta. 1998;1387:17–31. - PubMed
-
- Makin G, Dive C. Apoptosis and cancer chemotherapy. Trends Cell Biol. 2001;11:S22–S26. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

