Unusual features of pomoviral RNA movement
- PMID: 22203822
- PMCID: PMC3244614
- DOI: 10.3389/fmicb.2011.00259
Unusual features of pomoviral RNA movement
Abstract
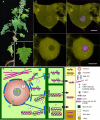
Potato mop-top pomovirus (PMTV) is one of a few viruses that can move systemically in plants in the absence of the capsid protein (CP). Pomoviruses encode the triple gene block genetic module of movement proteins (TGB 1, 2, and 3) and recent research suggests that PMTV RNA is transported either as ribonucleoprotein (RNP) complexes containing TGB1 or encapsidated in virions containing TGB1. Furthermore, there are different requirements for local or systemic (long-distance) movement. Research suggests that nucleolar passage of TGB1 may be important for the long-distance movement of both RNP and virions. Moreover, and uniquely, the long-distance movement of the CP-encoding RNA requires expression of both major and minor CP subunits and is inhibited when only the major CP sub unit is expressed. This paper reviews pomovirus research and presents a current model for RNA movement.
Keywords: BSBV; BVQ; PMTV; Pomovirus; TGB; microtubules; nucleolus; nucleus.
Figures

Similar articles
-
Unusual long-distance movement strategies of Potato mop-top virus RNAs in Nicotiana benthamiana.Mol Plant Microbe Interact. 2009 Apr;22(4):381-90. doi: 10.1094/MPMI-22-4-0381. Mol Plant Microbe Interact. 2009. PMID: 19271953
-
The N-terminal domain of PMTV TGB1 movement protein is required for nucleolar localization, microtubule association, and long-distance movement.Mol Plant Microbe Interact. 2010 Nov;23(11):1486-97. doi: 10.1094/MPMI-05-10-0105. Mol Plant Microbe Interact. 2010. PMID: 20923354
-
Importin-α-mediated nucleolar localization of potato mop-top virus TRIPLE GENE BLOCK1 (TGB1) protein facilitates virus systemic movement, whereas TGB1 self-interaction is required for cell-to-cell movement in Nicotiana benthamiana.Plant Physiol. 2015 Mar;167(3):738-52. doi: 10.1104/pp.114.254938. Epub 2015 Jan 9. Plant Physiol. 2015. PMID: 25576325 Free PMC article.
-
Varied movement strategies employed by triple gene block-encoding viruses.Mol Plant Microbe Interact. 2010 Oct;23(10):1231-47. doi: 10.1094/MPMI-04-10-0086. Mol Plant Microbe Interact. 2010. PMID: 20831404 Review.
-
Understanding the intracellular trafficking and intercellular transport of potexviruses in their host plants.Front Plant Sci. 2014 Mar 18;5:60. doi: 10.3389/fpls.2014.00060. eCollection 2014. Front Plant Sci. 2014. PMID: 24672528 Free PMC article. Review.
Cited by
-
The Coat Protein of Citrus Yellow Vein Clearing Virus Interacts with Viral Movement Proteins and Serves as an RNA Silencing Suppressor.Viruses. 2019 Apr 5;11(4):329. doi: 10.3390/v11040329. Viruses. 2019. PMID: 30959816 Free PMC article.
-
Small hydrophobic viral proteins involved in intercellular movement of diverse plant virus genomes.AIMS Microbiol. 2020 Sep 21;6(3):305-329. doi: 10.3934/microbiol.2020019. eCollection 2020. AIMS Microbiol. 2020. PMID: 33134746 Free PMC article. Review.
-
Deciphering the mechanism of defective interfering RNA (DI RNA) biogenesis reveals that a viral protein and the DI RNA act antagonistically in virus infection.J Virol. 2013 Jun;87(11):6091-103. doi: 10.1128/JVI.03322-12. Epub 2013 Mar 20. J Virol. 2013. PMID: 23514891 Free PMC article.
-
Viral and cellular factors involved in Phloem transport of plant viruses.Front Plant Sci. 2013 May 24;4:154. doi: 10.3389/fpls.2013.00154. eCollection 2013. Front Plant Sci. 2013. PMID: 23745125 Free PMC article.
-
Complete Genomic Characterization of Two Beet Soil-Borne Virus Isolates from Turkey: Implications of Comparative Analysis of Genome Sequences.Plant Pathol J. 2021 Apr;37(2):152-161. doi: 10.5423/PPJ.OA.12.2020.0223. Epub 2021 Apr 1. Plant Pathol J. 2021. PMID: 33866757 Free PMC article.
References
-
- Cowan G. H., Torrance L., Reavy B. (1997). Detection of potato mop-top virus capsid readthrough protein in virus particles. J. Gen. Virol. 78, 1779–1783 - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

