Adjustable passive stiffness in mouse bladder: regulated by Rho kinase and elevated following partial bladder outlet obstruction
- PMID: 22205227
- PMCID: PMC3330714
- DOI: 10.1152/ajprenal.00177.2011
Adjustable passive stiffness in mouse bladder: regulated by Rho kinase and elevated following partial bladder outlet obstruction
Abstract
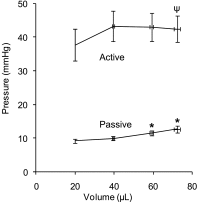
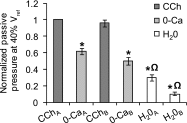
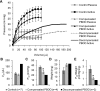
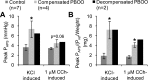
Detrusor smooth muscle (DSM) contributes to bladder wall tension during filling, and bladder wall deformation affects the signaling system that leads to urgency. The length-passive tension (L-T(p)) relationship in rabbit DSM can adapt with length changes over time and exhibits adjustable passive stiffness (APS) characterized by a L-T(p) curve that is a function of both activation and strain history. Muscle activation with KCl, carbachol (CCh), or prostaglandin E(2) at short muscle lengths can increase APS that is revealed by elevated pseudo-steady-state T(p) at longer lengths compared with prior T(p) measurements at those lengths, and APS generation is inhibited by the Rho Kinase (ROCK) inhibitor H-1152. In the current study, mouse bladder strips exhibited both KCl- and CCh-induced APS. Whole mouse bladders demonstrated APS which was measured as an increase in pressure during passive filling in calcium-free solution following CCh precontraction compared with pressure during filling without precontraction. In addition, CCh-induced APS in whole mouse bladder was inhibited by H-1152, indicating that ROCK activity may regulate bladder compliance during filling. Furthermore, APS in whole mouse bladder was elevated 2 wk after partial bladder outlet obstruction, suggesting that APS may be relevant in diseases affecting bladder mechanics. The presence of APS in mouse bladder will permit future studies of APS regulatory pathways and potential alterations of APS in disease models using knockout transgenetic mice.
Figures

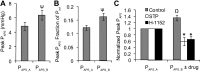
Similar articles
-
Rhythmic contraction generates adjustable passive stiffness in rabbit detrusor.J Appl Physiol (1985). 2010 Mar;108(3):544-53. doi: 10.1152/japplphysiol.01079.2009. Epub 2010 Jan 7. J Appl Physiol (1985). 2010. PMID: 20056849 Free PMC article.
-
Evidence that actomyosin cross bridges contribute to "passive" tension in detrusor smooth muscle.Am J Physiol Renal Physiol. 2010 Jun;298(6):F1424-35. doi: 10.1152/ajprenal.00635.2009. Epub 2010 Apr 7. Am J Physiol Renal Physiol. 2010. PMID: 20375119 Free PMC article.
-
Effects of fasudil, a Rho-kinase inhibitor, on contraction of pig bladder tissues with or without urothelium.Int J Urol. 2009 Dec;16(12):959-66. doi: 10.1111/j.1442-2042.2009.02397.x. Int J Urol. 2009. PMID: 19817915
-
Obstruction alters muscarinic receptor-coupled RhoA/Rho-kinase pathway in the urinary bladder of the rat.Neurourol Urodyn. 2009;28(3):257-62. doi: 10.1002/nau.20625. Neurourol Urodyn. 2009. PMID: 19229948
-
Age-dependent contribution of Rho kinase in carbachol-induced contraction of human detrusor smooth muscle in vitro.Acta Pharmacol Sin. 2014 Jan;35(1):74-81. doi: 10.1038/aps.2013.126. Epub 2013 Oct 14. Acta Pharmacol Sin. 2014. PMID: 24122009 Free PMC article.
Cited by
-
Comparative-fill urodynamics in individuals with and without detrusor overactivity supports a conceptual model for dynamic elasticity regulation.Neurourol Urodyn. 2020 Feb;39(2):707-714. doi: 10.1002/nau.24255. Epub 2019 Dec 19. Neurourol Urodyn. 2020. PMID: 31856359 Free PMC article.
-
High Hydrostatic Pressure Exacerbates Bladder Fibrosis through Activating Piezo1.Curr Med Sci. 2024 Aug;44(4):718-725. doi: 10.1007/s11596-024-2881-3. Epub 2024 Jun 27. Curr Med Sci. 2024. PMID: 38926331
-
Modeling the influence of acute changes in bladder elasticity on pressure and wall tension during filling.J Mech Behav Biomed Mater. 2017 Jul;71:192-200. doi: 10.1016/j.jmbbm.2017.02.020. Epub 2017 Feb 20. J Mech Behav Biomed Mater. 2017. PMID: 28343086 Free PMC article.
-
An external compress-release protocol induces dynamic elasticity in the porcine bladder: A novel technique for the treatment of overactive bladder?Neurourol Urodyn. 2019 Jun;38(5):1222-1228. doi: 10.1002/nau.23992. Epub 2019 Apr 4. Neurourol Urodyn. 2019. PMID: 30947371 Free PMC article.
-
Evidence for a common mechanism for spontaneous rhythmic contraction and myogenic contraction induced by quick stretch in detrusor smooth muscle.Physiol Rep. 2013 Nov;1(6):e00168. doi: 10.1002/phy2.168. Epub 2013 Nov 22. Physiol Rep. 2013. PMID: 24400167 Free PMC article.
References
-
- Almasri AM, Ratz PH, Speich JE. Length adaptation of the passive-to-active tension ratio in rabbit detrusor. Ann Biomed Eng 38: 2594–2605, 2010 - PubMed
-
- Austin JC, Chacko SK, DiSanto M, Canning DA, Zderic SA. A male murine model of partial bladder outlet obstruction reveals changes in detrusor morphology, contractility and myosin isoform expression. J Urol 172: 1524–1528, 2004 - PubMed
-
- Bai TR, Bates JH, Brusasco V, Camoretti-Mercado B, Chitano P, Deng LH, Dowell M, Fabry B, Ford LE, Fredberg JJ, Gerthoffer WT, Gilbert SH, Gunst SJ, Hai CM, Halayko AJ, Hirst SJ, James AL, Janssen LJ, Jones KA, King GG, Lakser OJ, Lambert RK, Lauzon AM, Lutchen KR, Maksym GN, Meiss RA, Mijailovich SM, Mitchell HW, Mitchell RW, Mitzner W, Murphy TM, Paré PD, Schellenberg RR, Seow CY, Sieck GC, Smith PG, Smolensky AV, Solway J, Stephens NL, Stewart AG, Tang DD, Wang L. On the terminology for describing the length-force relationship and its changes in airway smooth muscle. J Appl Physiol 97: 2029–2034, 2004 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

