Quantitative differences among EMG activities of muscles innervated by subpopulations of hypoglossal and upper spinal motoneurons during non-REM sleep - REM sleep transitions: a window on neural processes in the sleeping brain
- PMID: 22205596
- PMCID: PMC5222572
- DOI: 10.4449/aib.v149i4.1385
Quantitative differences among EMG activities of muscles innervated by subpopulations of hypoglossal and upper spinal motoneurons during non-REM sleep - REM sleep transitions: a window on neural processes in the sleeping brain
Abstract
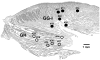
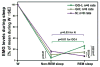
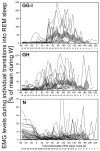
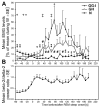
In the rat, a species widely used to study the neural mechanisms of sleep and motor control, lingual electromyographic activity (EMG) is minimal during non-rapid eye movement (non-REM) sleep and then phasic twitches gradually increase after the onset of REM sleep. To better characterize the central neural processes underlying this pattern, we quantified EMG of muscles innervated by distinct subpopulations of hypoglossal motoneurons and nuchal (N) EMG during transitions from non-REM sleep to REM sleep. In 8 chronically instrumented rats, we recorded cortical EEG, EMG at sites near the base of the tongue where genioglossal and intrinsic muscle fibers predominate (GG-I), EMG of the geniohyoid (GH) muscle, and N EMG. Sleep-wake states were identified and EMGs quantified relative to their mean levels in wakefulness in successive 10 s epochs. During non-REM sleep, the average EMG levels differed among the three muscles, with the order being N>GH>GG-I. During REM sleep, due to different magnitudes of phasic twitches, the order was reversed to GG-I>GH>N. GG-I and GH exhibited a gradual increase of twitching that peaked at 70-120 s after the onset of REM sleep and then declined if the REM sleep episode lasted longer. We propose that a common phasic excitatory generator impinges on motoneuron pools that innervate different muscles, but twitching magnitudes are different due to different levels of tonic motoneuronal hyperpolarization. We also propose that REM sleep episodes of average durations are terminated by intense activity of the central generator of phasic events, whereas long REM sleep episodes end as a result of a gradual waning of the tonic disfacilitatory and inhibitory processes.
Figures



References
-
- Aldes LD. Subcompartmental organization of the ventral (protrusor) compartment in the hypoglossal nucleus of the rat. J Comp Neurol. 1995;353:89–108. - PubMed
-
- Altschuler SM, Bao X, Miselis RR. Dendritic architecture of hypoglossal motoneurons projecting to extrinsic tongue musculature in the rat. J Comp Neurol. 1994;342:538–550. - PubMed
-
- Arnulf I. The ‘scanning hypothesis’ of rapid eye movements during REM sleep: a review of the evidence. Arch Ital Biol. 2011;151:367–382. (this issue) - PubMed
-
- Aserinsky E, Kleitman N. Regularly occurring periods of eye motility, and concomitant phenomena, during sleep. Science. 1953;118:273–274. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
