Infrared spectroscopy and microscopy in cancer research and diagnosis
- PMID: 22206042
- PMCID: PMC3236568
Infrared spectroscopy and microscopy in cancer research and diagnosis
Abstract
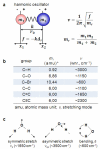
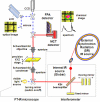
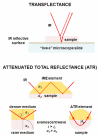
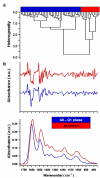
Since the middle of 20(th) century infrared (IR) spectroscopy coupled to microscopy (IR microspectroscopy) has been recognized as a non destructive, label free, highly sensitive and specific analytical method with many potential useful applications in different fields of biomedical research and in particular cancer research and diagnosis. Although many technological improvements have been made to facilitate biomedical applications of this powerful analytical technique, it has not yet properly come into the scientific background of many potential end users. Therefore, to achieve those fundamental objectives an interdisciplinary approach is needed with basic scientists, spectroscopists, biologists and clinicians who must effectively communicate and understand each other's requirements and challenges. In this review we aim at illustrating some principles of Fourier transform (FT) Infrared (IR) vibrational spectroscopy and microscopy (microFT-IR) as a useful method to interrogate molecules in specimen by mid-IR radiation. Penetrating into basics of molecular vibrations might help us to understand whether, when and how complementary information obtained by microFT-IR could become useful in our research and/or diagnostic activities. MicroFT-IR techniques allowing to acquire information about the molecular composition and structure of a sample within a micrometric scale in a matter of seconds will be illustrated as well as some limitations will be discussed. How biochemical, structural, and dynamical information about the systems can be obtained by bench top microFT-IR instrumentation will be also presented together with some methods to treat and interpret IR spectral data and applicative examples. The mid-IR absorbance spectrum is one of the most information-rich and concise way to represent the whole "… omics" of a cell and, as such, fits all the characteristics for the development of a clinically useful biomarker.
Keywords: Molecular vibrations; cancer biomarker; cancer diagnosis; infrared microspectroscopy; infrared radiation; mid-infrared absorbance spectroscopy; pre-clinical drug screening; synchrotron radiation; unsupervised multivariate analysis; vibrational spectroscopy.
Figures

References
LinkOut - more resources
Full Text Sources
Other Literature Sources
