Riluzole and gabapentinoids activate glutamate transporters to facilitate glutamate-induced glutamate release from cultured astrocytes
- PMID: 22206816
- PMCID: PMC3268950
- DOI: 10.1016/j.ejphar.2011.12.015
Riluzole and gabapentinoids activate glutamate transporters to facilitate glutamate-induced glutamate release from cultured astrocytes
Abstract
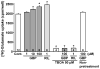
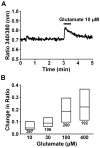
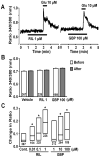
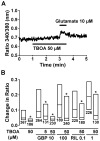
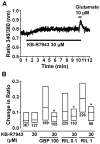
We have recently demonstrated that the glutamate transporter activator riluzole paradoxically enhanced glutamate-induced glutamate release from cultured astrocytes. We further showed that both riluzole and the α(2)δ subunit ligand gabapentin activated descending inhibition in rats by increasing glutamate receptor signaling in the locus coeruleus and hypothesized that these drugs share common mechanisms to enhance glutamate release from astrocytes. In the present study, we examined the effects of riluzole and gabapentin on glutamate uptake and release and glutamate-induced Ca(2+) responses in primary cultures of astrocytes. Riluzole and gabapentin facilitated glutamate-induced glutamate release from astrocytes and significantly increased glutamate uptake, the latter being completely blocked by the non-selective glutamate transporter blocker DL-threo-β-benzyloxyaspartic acid (DL-TBOA). Riluzole and gabapentin also enhanced the glutamate-induced increase in intracellular Ca(2+) concentrations. Some α(2)δ subunit ligands, pregabalin and L-isoleucine, enhanced the glutamate-induced Ca(2+) response, whereas another, 3-exo-aminobicyclo[2.2.1]heptane-2-exo-carboxylic acid (ABHCA), did not. The enhancement of glutamate-induced intracellular Ca(2+) response by riluzole and gabapentin was blocked by the DL-TBOA and an inhibitor of Na(+)/Ca(2+) exchange, 2-[2-[4-(4-nitrobenzyloxy)phenyl]ethyl]isothiurea (KB-R7943). Gabapentin's enhancement of Ca(2+) increase was specific to glutamate stimulation, as it was not mimicked with stimulation by ATP. These results suggest that riluzole and gabapentin enhance Na(+)-glutamate co-transport through glutamate transporters, induce subsequent Ca(2+) influx via the reverse mode of Na(+)/Ca(2+) exchange, and thereby facilitate Ca(2+)-dependent glutamate release by glutamate in astrocytes. The present study also demonstrates a novel target of gabapentinoid action in astrocytes other than α(2)δ subunits in neurons.
Copyright © 2011 Elsevier B.V. All rights reserved.
Figures
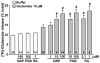

Similar articles
-
Gabapentin increases extracellular glutamatergic level in the locus coeruleus via astroglial glutamate transporter-dependent mechanisms.Neuropharmacology. 2014 Jun;81:95-100. doi: 10.1016/j.neuropharm.2014.01.040. Epub 2014 Feb 1. Neuropharmacology. 2014. PMID: 24495399 Free PMC article.
-
Activation of glutamate transporters in the locus coeruleus paradoxically activates descending inhibition in rats.Brain Res. 2010 Mar 4;1317:80-6. doi: 10.1016/j.brainres.2009.12.086. Epub 2010 Jan 6. Brain Res. 2010. PMID: 20059984 Free PMC article.
-
Effects of gabapentin and pregabalin on K+-evoked 3H-GABA and 3H-glutamate release from human neocortical synaptosomes.Naunyn Schmiedebergs Arch Pharmacol. 2009 Apr;379(4):361-9. doi: 10.1007/s00210-008-0370-z. Epub 2008 Nov 11. Naunyn Schmiedebergs Arch Pharmacol. 2009. PMID: 19002437
-
Acute modulation of calcium currents and synaptic transmission by gabapentinoids.Channels (Austin). 2010 Nov-Dec;4(6):490-6. doi: 10.4161/chan.4.6.12864. Epub 2010 Nov 1. Channels (Austin). 2010. PMID: 21150315 Review.
-
Sodium-calcium exchanger modulates the L-glutamate Ca(i) (2+) signalling in type-1 cerebellar astrocytes.Adv Exp Med Biol. 2013;961:267-74. doi: 10.1007/978-1-4614-4756-6_22. Adv Exp Med Biol. 2013. PMID: 23224886 Review.
Cited by
-
Ketamine Within Clinically Effective Range Inhibits Glutamate Transmission From Astrocytes to Neurons and Disrupts Synchronization of Astrocytic SICs.Front Cell Neurosci. 2019 Jun 11;13:240. doi: 10.3389/fncel.2019.00240. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31244607 Free PMC article.
-
Targeting glia cells: novel perspectives for the treatment of neuropsychiatric diseases.Curr Neuropharmacol. 2013 Mar;11(2):171-85. doi: 10.2174/1570159X11311020004. Curr Neuropharmacol. 2013. PMID: 23997752 Free PMC article.
-
Inflammation Models of Depression in Rodents: Relevance to Psychotropic Drug Discovery.Int J Neuropsychopharmacol. 2016 Sep 21;19(9):pyw028. doi: 10.1093/ijnp/pyw028. Print 2016 Sep. Int J Neuropsychopharmacol. 2016. PMID: 27026361 Free PMC article. Review.
-
Effect of glutamate and riluzole on manganese-induced apoptotic cell signaling in neuronally differentiated mouse P19 Cells.Neurochem Int. 2012 Jul;61(1):25-33. doi: 10.1016/j.neuint.2012.04.015. Epub 2012 Apr 21. Neurochem Int. 2012. PMID: 22543103 Free PMC article.
-
Gabapentin increases extracellular glutamatergic level in the locus coeruleus via astroglial glutamate transporter-dependent mechanisms.Neuropharmacology. 2014 Jun;81:95-100. doi: 10.1016/j.neuropharm.2014.01.040. Epub 2014 Feb 1. Neuropharmacology. 2014. PMID: 24495399 Free PMC article.
References
-
- Anderson CM, Swanson RA. Astrocyte glutamate transport: review of properties, regulation, and physiological functions. Glia. 2000;32:1–14. - PubMed
-
- Bertrand S, Nouel D, Morin F, Nagy F, Lacaille JC. Gabapentin actions on Kir3 currents and N-type Ca2+ channels via GABAB receptors in hippocampal pyramidal cells. Synapse. 2003;50:95–109. - PubMed
-
- Brooks BR. Managing amyotrophic lateral sclerosis: slowing disease progression and improving patient quality of life. Ann Neurol. 2009;65(Suppl 1):S17–23. - PubMed
-
- Coderre TJ, Kumar N, Lefebvre CD, Yu JS. A comparison of the glutamate release inhibition and anti-allodynic effects of gabapentin, lamotrigine, and riluzole in a model of neuropathic pain. J Neurochem. 2007;100:1289–1299. - PubMed
-
- Davies A, Hendrich J, Van Minh AT, Wratten J, Douglas L, Dolphin AC. Functional biology of the alpha(2)delta subunits of voltage-gated calcium channels. Trends Pharmacol Sci. 2007;28:220–228. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

