High pressure ices
- PMID: 22207625
- PMCID: PMC3271906
- DOI: 10.1073/pnas.1118694109
High pressure ices
Abstract
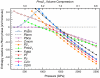
H(2)O will be more resistant to metallization than previously thought. From computational evolutionary structure searches, we find a sequence of new stable and meta-stable structures for the ground state of ice in the 1-5 TPa (10 to 50 Mbar) regime, in the static approximation. The previously proposed Pbcm structure is superseded by a Pmc2(1) phase at p = 930 GPa, followed by a predicted transition to a P2(1) crystal structure at p = 1.3 TPa. This phase, featuring higher coordination at O and H, is stable over a wide pressure range, reaching 4.8 TPa. We analyze carefully the geometrical changes in the calculated structures, especially the buckling at the H in O-H-O motifs. All structures are insulating--chemistry burns a deep and (with pressure increase) lasting hole in the density of states near the highest occupied electronic levels of what might be component metallic lattices. Metallization of ice in our calculations occurs only near 4.8 TPa, where the metallic C2/m phase becomes most stable. In this regime, zero-point energies much larger than typical enthalpy differences suggest possible melting of the H sublattice, or even the entire crystal.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
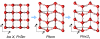
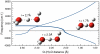
 ), Pbcm, and Pmc21, seen along the a axis. Unit cells are indicated (
), Pbcm, and Pmc21, seen along the a axis. Unit cells are indicated ( cell for ice X). All structures are at p = 700 GPa (and ground-states).
cell for ice X). All structures are at p = 700 GPa (and ground-states).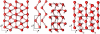
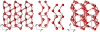
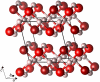
 structure. Left: along the c axis; middle: the two sublattices, along the a axis; right: one of the two interpenetrating networks.
structure. Left: along the c axis; middle: the two sublattices, along the a axis; right: one of the two interpenetrating networks.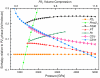
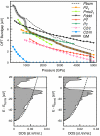
Similar articles
-
Lithium amide (LiNH2) under pressure.J Phys Chem A. 2012 Oct 11;116(40):10027-36. doi: 10.1021/jp3078387. Epub 2012 Oct 2. J Phys Chem A. 2012. PMID: 22978817
-
Decomposition and Terapascal phases of water ice.Phys Rev Lett. 2013 Jun 14;110(24):245701. doi: 10.1103/PhysRevLett.110.245701. Epub 2013 Jun 11. Phys Rev Lett. 2013. PMID: 25165937
-
New metallic ice phase under high pressure.Phys Chem Chem Phys. 2024 Nov 7;26(43):27783-27790. doi: 10.1039/d4cp02543a. Phys Chem Chem Phys. 2024. PMID: 39470353
-
Quantum simulation of thermally-driven phase transition and oxygen K-edge x-ray absorption of high-pressure ice.Sci Rep. 2013 Nov 20;3:3272. doi: 10.1038/srep03272. Sci Rep. 2013. PMID: 24253589 Free PMC article.
-
Molecular origins of homogeneous crystal nucleation.Annu Rev Chem Biomol Eng. 2012;3:157-82. doi: 10.1146/annurev-chembioeng-062011-081029. Epub 2012 Mar 9. Annu Rev Chem Biomol Eng. 2012. PMID: 22468601 Review.
Cited by
-
Thermal expansion anomaly regulated by entropy.Sci Rep. 2014 Nov 13;4:7043. doi: 10.1038/srep07043. Sci Rep. 2014. PMID: 25391631 Free PMC article.
-
Room temperature electrofreezing of water yields a missing dense ice phase in the phase diagram.Nat Commun. 2019 Apr 26;10(1):1925. doi: 10.1038/s41467-019-09950-z. Nat Commun. 2019. PMID: 31028288 Free PMC article.
-
Neutron diffraction observations of interstitial protons in dense ice.Proc Natl Acad Sci U S A. 2013 Jun 25;110(26):10552-6. doi: 10.1073/pnas.1309277110. Epub 2013 Jun 11. Proc Natl Acad Sci U S A. 2013. PMID: 23757495 Free PMC article.
-
Observation of plastic ice VII by quasi-elastic neutron scattering.Nature. 2025 Apr;640(8059):662-667. doi: 10.1038/s41586-025-08750-4. Epub 2025 Feb 12. Nature. 2025. PMID: 39938568 Free PMC article.
-
Stabilization of ammonia-rich hydrate inside icy planets.Proc Natl Acad Sci U S A. 2017 Aug 22;114(34):9003-9008. doi: 10.1073/pnas.1706244114. Epub 2017 Aug 7. Proc Natl Acad Sci U S A. 2017. PMID: 28784809 Free PMC article.
References
-
- Petrenko VF, Whitworth RW. Physics of Ice. Oxford: Oxford University Press; 1999.
-
- Salzmann CG, Radaelli PG, Mayer E, Finney JL. Ice XV: A New Thermodynamically Stable Phase of Ice. Phys Rev Lett. 2009;103:105701. - PubMed
-
- Ross M. The ice layer in Uranus and Neptune—diamonds in the sky? Nature. 1981;292:435–436.
-
- Podolak M, Weizman A, Marley M. Comparative models of Uranus and Neptune. Planet Space Sci. 1995;43:1517–1522.
-
- Beaulieu J-P, et al. Discovery of a cool planet of 5.5 Earth masses through gravitational microlensing. Nature. 2006;439:437–440. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

