Ancient Pbx-Hox signatures define hundreds of vertebrate developmental enhancers
- PMID: 22208168
- PMCID: PMC3261376
- DOI: 10.1186/1471-2164-12-637
Ancient Pbx-Hox signatures define hundreds of vertebrate developmental enhancers
Abstract
Background: Gene regulation through cis-regulatory elements plays a crucial role in development and disease. A major aim of the post-genomic era is to be able to read the function of cis-regulatory elements through scrutiny of their DNA sequence. Whilst comparative genomics approaches have identified thousands of putative regulatory elements, our knowledge of their mechanism of action is poor and very little progress has been made in systematically de-coding them.
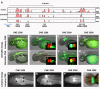
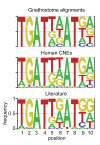
Results: Here, we identify ancient functional signatures within vertebrate conserved non-coding elements (CNEs) through a combination of phylogenetic footprinting and functional assay, using genomic sequence from the sea lamprey as a reference. We uncover a striking enrichment within vertebrate CNEs for conserved binding-site motifs of the Pbx-Hox hetero-dimer. We further show that these predict reporter gene expression in a segment specific manner in the hindbrain and pharyngeal arches during zebrafish development.
Conclusions: These findings evoke an evolutionary scenario in which many CNEs evolved early in the vertebrate lineage to co-ordinate Hox-dependent gene-regulatory interactions that pattern the vertebrate head. In a broader context, our evolutionary analyses reveal that CNEs are composed of tightly linked transcription-factor binding-sites (TFBSs), which can be systematically identified through phylogenetic footprinting approaches. By placing a large number of ancient vertebrate CNEs into a developmental context, our findings promise to have a significant impact on efforts toward de-coding gene-regulatory elements that underlie vertebrate development, and will facilitate building general models of regulatory element evolution.
Figures


Similar articles
-
A Simple Predictive Enhancer Syntax for Hindbrain Patterning Is Conserved in Vertebrate Genomes.PLoS One. 2015 Jul 1;10(7):e0130413. doi: 10.1371/journal.pone.0130413. eCollection 2015. PLoS One. 2015. PMID: 26131856 Free PMC article.
-
Analysis of lamprey meis genes reveals that conserved inputs from Hox, Meis and Pbx proteins control their expression in the hindbrain and neural tube.Dev Biol. 2021 Nov;479:61-76. doi: 10.1016/j.ydbio.2021.07.014. Epub 2021 Jul 24. Dev Biol. 2021. PMID: 34310923
-
A Hox regulatory network of hindbrain segmentation is conserved to the base of vertebrates.Nature. 2014 Oct 23;514(7523):490-3. doi: 10.1038/nature13723. Epub 2014 Sep 14. Nature. 2014. PMID: 25219855 Free PMC article.
-
The vertebrate Hox gene regulatory network for hindbrain segmentation: Evolution and diversification: Coupling of a Hox gene regulatory network to hindbrain segmentation is an ancient trait originating at the base of vertebrates.Bioessays. 2016 Jun;38(6):526-38. doi: 10.1002/bies.201600010. Epub 2016 Mar 29. Bioessays. 2016. PMID: 27027928 Review.
-
A Hox gene regulatory network for hindbrain segmentation.Curr Top Dev Biol. 2020;139:169-203. doi: 10.1016/bs.ctdb.2020.03.001. Epub 2020 Apr 9. Curr Top Dev Biol. 2020. PMID: 32450960 Review.
Cited by
-
A unique stylopod patterning mechanism by Shox2-controlled osteogenesis.Development. 2016 Jul 15;143(14):2548-60. doi: 10.1242/dev.138750. Epub 2016 Jun 10. Development. 2016. PMID: 27287812 Free PMC article.
-
Purifying selection in deeply conserved human enhancers is more consistent than in coding sequences.PLoS One. 2014 Jul 25;9(7):e103357. doi: 10.1371/journal.pone.0103357. eCollection 2014. PLoS One. 2014. PMID: 25062004 Free PMC article.
-
Evolution of the expression and regulation of the nuclear hormone receptor ERR gene family in the chordate lineage.Dev Biol. 2023 Dec;504:12-24. doi: 10.1016/j.ydbio.2023.09.003. Epub 2023 Sep 9. Dev Biol. 2023. PMID: 37696353 Free PMC article.
-
I-SceI Meganuclease-mediated transgenesis in the acorn worm, Saccoglossus kowalevskii.Dev Biol. 2019 Jan 1;445(1):8-15. doi: 10.1016/j.ydbio.2018.10.022. Epub 2018 Nov 6. Dev Biol. 2019. PMID: 30412702 Free PMC article.
-
A Simple Predictive Enhancer Syntax for Hindbrain Patterning Is Conserved in Vertebrate Genomes.PLoS One. 2015 Jul 1;10(7):e0130413. doi: 10.1371/journal.pone.0130413. eCollection 2015. PLoS One. 2015. PMID: 26131856 Free PMC article.
References
-
- Lettice LA, Heaney SJ, Purdie LA, Li L, de Beer P, Oostra BA, Goode D, Elgar G, Hill RE, de Graaff E. A long-range Shh enhancer regulates expression in the developing limb and fin and is associated with preaxial polydactyly. Hum Mol Genet. 2003;12:1725–35. - PubMed
-
- Siepel A, Bejerano G, Pedersen JS, Hinrichs AS, Hou M, Rosenbloom K, Clawson H, Spieth J, Hillier LW, Richards S, Weinstock GM, Wilson RK, Gibbs RA, Kent WJ, Miller W, Haussler D. Evolutionarily conserved elements in vertebrate, insect, worm, and yeast genomes. Genome Res. 2005;15:1034–50. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

