White-matter astrocytes, axonal energy metabolism, and axonal degeneration in multiple sclerosis
- PMID: 22214904
- PMCID: PMC3293127
- DOI: 10.1038/jcbfm.2011.193
White-matter astrocytes, axonal energy metabolism, and axonal degeneration in multiple sclerosis
Abstract
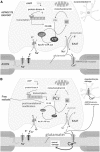
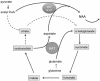
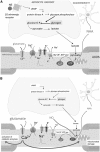
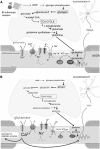
In patients with multiple sclerosis (MS), a diffuse axonal degeneration occurring throughout the white matter of the central nervous system causes progressive neurologic disability. The underlying mechanism is unclear. This review describes a number of pathways by which dysfunctional astrocytes in MS might lead to axonal degeneration. White-matter astrocytes in MS show a reduced metabolism of adenosine triphosphate-generating phosphocreatine, which may impair the astrocytic sodium potassium pump and lead to a reduced sodium-dependent glutamate uptake. Astrocytes in MS white matter appear to be deficient in β(2) adrenergic receptors, which are involved in stimulating glycogenolysis and suppressing inducible nitric oxide synthase (NOS2). Glutamate toxicity, reduced astrocytic glycogenolysis leading to reduced lactate and glutamine production, and enhanced nitric oxide (NO) levels may all impair axonal mitochondrial metabolism, leading to axonal degeneration. In addition, glutamate-mediated oligodendrocyte damage and impaired myelination caused by a decreased production of N-acetylaspartate by axonal mitochondria might also contribute to axonal loss. White-matter astrocytes may be considered as a potential target for neuroprotective MS therapies.
Figures
References
-
- Aksenov M, Aksenova M, Butterfield DA, Markesbery WR. Oxidative modification of creatine kinase BB in Alzheimer's disease brain. J Neurochem. 2000;74:2520–2527. - PubMed
-
- Aksenova MV, Aksenov MY, Payne RM, Trojanowski JQ, Schmidt ML, Carney JM, Butterfield DA, Markesbery WR. Oxidation of cytosolic proteins and expression of creatine kinase BB in frontal lobe in different neurodegenerative disorders. Dement Geriatr Cogn Disord. 1999;10:158–165. - PubMed
-
- Alix JJ, Domingues AM. White matter synapses: form, function, and dysfunction. Neurology. 2011;76:397–404. - PubMed
-
- Allaman I, Fiumelli H, Magistretti PJ, Martin JL. Fluoxetine regulates the expression of neurotrophic/growth factors and glucose metabolism in astrocytes. Psychopharmacology. 2011;216:75–84. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

