Prion uptake in the gut: identification of the first uptake and replication sites
- PMID: 22216002
- PMCID: PMC3245311
- DOI: 10.1371/journal.ppat.1002449
Prion uptake in the gut: identification of the first uptake and replication sites
Abstract
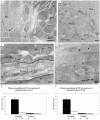
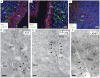
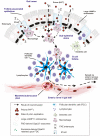
After oral exposure, prions are thought to enter Peyer's patches via M cells and accumulate first upon follicular dendritic cells (FDCs) before spreading to the nervous system. How prions are actually initially acquired from the gut lumen is not known. Using high-resolution immunofluorescence and cryo-immunogold electron microscopy, we report the trafficking of the prion protein (PrP) toward Peyer's patches of wild-type and PrP-deficient mice. PrP was transiently detectable at 1 day post feeding (dpf) within large multivesicular LAMP1-positive endosomes of enterocytes in the follicle-associated epithelium (FAE) and at much lower levels within M cells. Subsequently, PrP was detected on vesicles in the late endosomal compartments of macrophages in the subepithelial dome. At 7-21 dpf, increased PrP labelling was observed on the plasma membranes of FDCs in germinal centres of Peyer's patches from wild-type mice only, identifying FDCs as the first sites of PrP conversion and replication. Detection of PrP on extracellular vesicles displaying FAE enterocyte-derived A33 protein implied transport towards FDCs in association with FAE-derived vesicles. By 21 dpf, PrP was observed on the plasma membranes of neurons within neighbouring myenteric plexi. Together, these data identify a novel potential M cell-independent mechanism for prion transport, mediated by FAE enterocytes, which acts to initiate conversion and replication upon FDCs and subsequent infection of enteric nerves.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
Oral Prion Neuroinvasion Occurs Independently of PrPC Expression in the Gut Epithelium.J Virol. 2018 Sep 12;92(19):e01010-18. doi: 10.1128/JVI.01010-18. Print 2018 Oct 1. J Virol. 2018. PMID: 30021891 Free PMC article.
-
Disease-associated PrP in the enteric nervous system of scrapie-affected Suffolk sheep.J Gen Virol. 2003 May;84(Pt 5):1327-1338. doi: 10.1099/vir.0.18874-0. J Gen Virol. 2003. PMID: 12692300
-
M cell-depletion blocks oral prion disease pathogenesis.Mucosal Immunol. 2012 Mar;5(2):216-25. doi: 10.1038/mi.2011.68. Epub 2012 Feb 1. Mucosal Immunol. 2012. PMID: 22294048 Free PMC article.
-
Prion diseases and the gastrointestinal tract.Can J Gastroenterol. 2006 Jan;20(1):18-24. doi: 10.1155/2006/184528. Can J Gastroenterol. 2006. PMID: 16432555 Free PMC article. Review.
-
Dendritic cells and oral transmission of prion diseases.Adv Drug Deliv Rev. 2004 Apr 19;56(6):901-13. doi: 10.1016/j.addr.2003.09.006. Adv Drug Deliv Rev. 2004. PMID: 15063597 Review.
Cited by
-
Rapid transepithelial transport of prions following inhalation.J Virol. 2012 Dec;86(23):12731-40. doi: 10.1128/JVI.01930-12. Epub 2012 Sep 12. J Virol. 2012. PMID: 22973025 Free PMC article.
-
The role of microglia in prion diseases and possible therapeutic targets: a literature review.Prion. 2021 Dec;15(1):191-206. doi: 10.1080/19336896.2021.1991771. Prion. 2021. PMID: 34751640 Free PMC article. Review.
-
Cellular mechanisms responsible for cell-to-cell spreading of prions.Cell Mol Life Sci. 2018 Jul;75(14):2557-2574. doi: 10.1007/s00018-018-2823-y. Epub 2018 May 14. Cell Mol Life Sci. 2018. PMID: 29761205 Free PMC article. Review.
-
Editorial: Immunological consequences of antigen sampling at mucosal surfaces, volume II.Front Immunol. 2024 Oct 11;15:1499174. doi: 10.3389/fimmu.2024.1499174. eCollection 2024. Front Immunol. 2024. PMID: 39464893 Free PMC article. No abstract available.
-
Innervation and nerve-immune cell contacts in mouse Peyer's patches.Histol Histopathol. 2020 Apr;35(4):371-383. doi: 10.14670/HH-18-158. Epub 2019 Sep 5. Histol Histopathol. 2020. PMID: 31486062
References
-
- Prusiner SB. In: Fields Virology. Knipe DM, Howley PM, editors. Philadelphia: Lippincott Williams & Wilkins; 2007. pp. 3359–3091.
-
- Blattler T, Brandner S, Raeber AJ, Klein MA, Voigtlander T, et al. PrP-expressing tissue required for transfer of scrapie infectivity from spleen to brain. Nature. 1997;389:69–73. - PubMed
-
- Brandner S, Isenmann S, Raeber A, Fischer M, Sailer A, et al. Normal host prion protein necessary for scrapie-induced neurotoxicity. Nature. 1996;379:339–43. - PubMed
-
- Bueler H, Aguzzi A, Sailer A, Greiner RA, Autenried P, et al. Mice devoid of PrP are resistant to scrapie. Cell. 1993;73:1339–47. - PubMed
-
- Peretz D, Williamson RA, Matsunaga Y, Serban H, Pinilla C, et al. A conformational transition at the N terminus of the prion protein features in formation of the scrapie isoform. J Mol Biol. 1997;273:614–22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- BB/D00831X/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BBS/E/D/05241339/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BBS/E/A/00001660/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BBS/E/A/00001659/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/G003947/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

