A deep insight into the sialotranscriptome of the gulf coast tick, Amblyomma maculatum
- PMID: 22216098
- PMCID: PMC3244413
- DOI: 10.1371/journal.pone.0028525
A deep insight into the sialotranscriptome of the gulf coast tick, Amblyomma maculatum
Abstract
Background: Saliva of blood sucking arthropods contains compounds that antagonize their hosts' hemostasis, which include platelet aggregation, vasoconstriction and blood clotting; saliva of these organisms also has anti-inflammatory and immunomodullatory properties. Perhaps because hosts mount an active immune response against these compounds, the diversity of these compounds is large even among related blood sucking species. Because of these properties, saliva helps blood feeding as well as help the establishment of pathogens that can be transmitted during blood feeding.
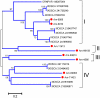
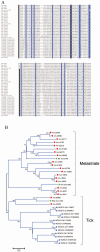
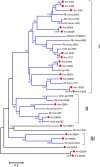
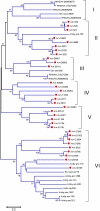
Methodology/principal findings: We have obtained 1,626,969 reads by pyrosequencing a salivary gland cDNA library from adult females Amblyomma maculatum ticks at different times of feeding. Assembly of this data produced 72,441 sequences larger than 149 nucleotides from which 15,914 coding sequences were extracted. Of these, 5,353 had >75% coverage to their best match in the non-redundant database from the National Center for Biotechnology information, allowing for the deposition of 4,850 sequences to GenBank. The annotated data sets are available as hyperlinked spreadsheets. Putative secreted proteins were classified in 133 families, most of which have no known function.
Conclusions/significance: This data set of proteins constitutes a mining platform for novel pharmacologically active proteins and for uncovering vaccine targets against A. maculatum and the diseases they carry.
Conflict of interest statement
Figures
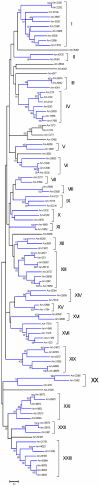
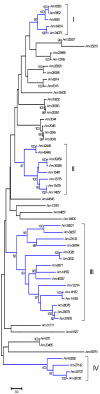

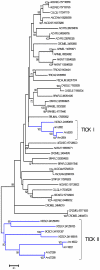
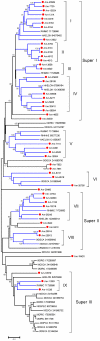

References
-
- Jones LD, Matthewson M, Nuttall PA. Saliva-activated transmission (SAT) of Thogoto virus: dynamics of SAT factor activity in the salivary glands of Rhipicephalus appendiculatus, Amblyomma variegatum, and Boophilus microplus ticks. Exp Appl Acarol. 1992;13:241–248. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

