Influenza vaccination for immunocompromised patients: systematic review and meta-analysis from a public health policy perspective
- PMID: 22216224
- PMCID: PMC3245259
- DOI: 10.1371/journal.pone.0029249
Influenza vaccination for immunocompromised patients: systematic review and meta-analysis from a public health policy perspective
Abstract
Background: Immunocompromised patients are vulnerable to severe or complicated influenza infection. Vaccination is widely recommended for this group. This systematic review and meta-analysis assesses influenza vaccination for immunocompromised patients in terms of preventing influenza-like illness and laboratory confirmed influenza, serological response and adverse events.
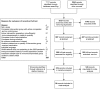
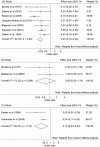
Methodology/principal findings: Electronic databases and grey literature were searched and records were screened against eligibility criteria. Data extraction and risk of bias assessments were performed in duplicate. Results were synthesised narratively and meta-analyses were conducted where feasible. Heterogeneity was assessed using I(2) and publication bias was assessed using Begg's funnel plot and Egger's regression test. Many of the 209 eligible studies included an unclear or high risk of bias. Meta-analyses showed a significant effect of preventing influenza-like illness (odds ratio [OR]=0.23; 95% confidence interval [CI]=0.16-0.34; p<0.001) and laboratory confirmed influenza infection (OR=0.15; 95% CI=0.03-0.63; p=0.01) through vaccinating immunocompromised patie nts compared to placebo or unvaccinated controls. We found no difference in the odds of influenza-like illness compared to vaccinated immunocompetent controls. The pooled odds of seroconversion were lower in vaccinated patients compared to immunocompetent controls for seasonal influenza A(H1N1), A(H3N2) and B. A similar trend was identified for seroprotection. Meta-analyses of seroconversion showed higher odds in vaccinated patients compared to placebo or unvaccinated controls, although this reached significance for influenza B only. Publication bias was not detected and narrative synthesis supported our findings. No consistent evidence of safety concerns was identified.
Conclusions/significance: Infection prevention and control strategies should recommend vaccinating immunocompromised patients. Potential for bias and confounding and the presence of heterogeneity mean the evidence reviewed is generally weak, although the directions of effects are consistent. Areas for further research are identified.
© 2011 Beck et al.
Conflict of interest statement
Figures
References
-
- Thompson WW, Shay DK, Weintraub E, Brammer L, Bridges CB, et al. Influenza-associated hospitalizations in the United States. JAMA. 2004;292:1333–1340. - PubMed
-
- Thompson WW, Shay DK, Weintraub E, Brammer L, Cox N, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA. 2003;289:179–186. - PubMed

